Advertisements
Advertisements
Questions
Find the number of atoms in the fcc unit cell.
Calculate the number of particles per unit cell in a face-centred cubic system.
In a face centred arrangement of atoms of an element, what will be the number of atoms present in respective unit cells?
Advertisements
Solution
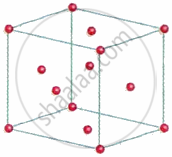
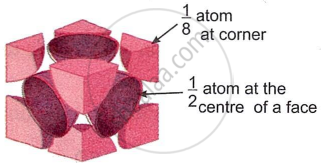
A face-centred cubic (fcc) unit cell has particles at the eight corners plus particles at the centre of its six faces.
Each particle present at the corner of a given unit cell is shared with seven other neighbouring unit cells. As a result, its contribution to the given unit cell is only `1/8`.
Thus, the number of particles present at corners per unit cell
= 8 corner atoms × `1/8` atom per unit cell = 1
Each particle at the centre of the six faces is shared with one neighbouring cube. Thus, 1/2 of each face particle belongs to the given unit cell. Thus, the number of particles present at faces per unit cell
= 6 atoms at the faces × `1/2` atom per unit cell = 3
Therefore, fcc unit cell has one corner particle plus 3 face particles, making a total of 4 particles per unit cell.
APPEARS IN
RELATED QUESTIONS
Write the relationship between radius of atom and edge length of fcc unit cell.
Calculate the number of unit cells in 0.3 g of a species having density of 8.5 g/cm3 and unit cell edge length 3.25 × 10-8 cm.
Derive the relationship between density of substance, its molar mass, and the unit cell edge length. Explain how you will calculate the number of particles, and a number of unit cells in x g of metal.
The pyranose structure of glucose is ____________.
What is the percentage of unoccupied space in fcc unit cell?
In bcc unit cell, the edge length (a) and radius of sphere (r) are related to each other by equation:
How many total constituent particles are present in simple cubic unit cell?
The number of atoms in 500 g of a fcc crystal of a metal with density d = 10 g/cm3 and cell edge 100 pm, is equal to ____________.
A metal crystallises in bcc unit cell with edge length 'a'. What will be the volume of one atom?
If the edge of a body-centred unit cell is 360 pm, what will be the approximate radius of the atom present in it? (in pm)
Sodium crystallizes in bcc structure with radius 1.86 × 10−8 cm. What is the length of unit cell of sodium?
An element crystallizes bcc type of unit cell, the density and edge length of unit cell is 4 g cm−3 and 500 pm respectively. What is the atomic mass of an element?
The coordination number of atoms in body-centred cubic structure (bcc) is ______.
Gold crystallises into face-centred cubic cells. The edge length of a unit cell is 4.08 × 10–8 cm. Calculate the density of gold. [Molar mass of gold = 197 g mol–1]
An element has a bee structure with unit cell edge length of 288 pm. How many unit cells and number of atoms are present in 200 g of the element?
In face centred cubic unit cell, what is the volume occupied?
Calculate the density of metal with molar mass 56 g mol- 1 that crystallises to form a bcc structure with edge length 288 pm.
An element with molar mass 2.7 × 10-2 kg/mol. Forms a cubic units cell with edge length of 405 pm. If the density is 2.7 × 103 kg/m3. Find the nature of a cubic unit cell.
What is the density of potassium, if it has a bcc structure with edge length 4Å?
(Atomic mass of K = 39)
What is base centred (or end-centred) unit cell?
The correct sequence of the atomic layers in cubic close packing is ______.
What would be the empirical formula of a compound having a unit cell containing A ion shared equally at the corner of the cube and B ion on the centre of faces of the cube?
An element crystallises in fee structure. If molar mass of element is 72.7 g mol -1, the mass of its one unit cell will be ______.
Which of the following metals exhibits minimum packing efficiency in its cubic system?
Calculate the molar mass of an element having a density of 19.2 g cm−3 if it forms an fcc structure [a3 × NA = 40 cm3 mol−1].
