Advertisements
Advertisements
Question
Explain with examples the term oxidation potential.
Explain
Advertisements
Solution
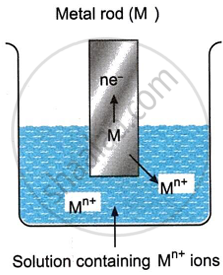
When a metal atom M present on the electrode loses n electrons, it gets oxidised into Mn+ ion as shown below.
\[\ce{M_{(s)} -> M{^{n+}_{(aq)}} + ne-}\]
The resulting ion, Mn+, enters the solution, and the electrons released during the oxidation process gather at the electrode. The procedure keeps going until the solution’s built-up charge prevents more action. Consequently, a negative potential is developed in the electrode. The resulting potential is known as an oxidation potential, and this kind of electrode assembly is referred to as an oxidation electrode.
shaalaa.com
Is there an error in this question or solution?
