Advertisements
Advertisements
Question
Explain the term ‘Interconversion of matter’. With reference to ice, water, and water vapour show diagrammatically the change of state of matter from solid to liquid to gaseous and back to the original state.
Advertisements
Solution
INTERCONVERSION OF MATTER:
“Change of state of matter from one state to another state and back to its original state is called inter-conversion of matter.”
The diagram showing the Change of State of Matter:

(a) ICE TO WATER (MELTING): Conversion of a solid into a liquid on heating
e.g. ice to water.
(b) WATER TO WATER VAPOUR (VAPORISATION): Conversion of a – liquid into vapours (or gas) on heating
e.g. water to water vapour.
(c) WATER VAPOUR TO WATER (LIQUEFACTION or CONDENSATION): Conversion of VAPOUR (or gas) into a LIQUID on cooling
e.g. water vapour to water.
(d) WATER TO ICE (SOLIDIFICATION OR FREEZING): Conversion of a LIQUID into a SOLID
e.g. water to ice.
RELATED QUESTIONS
What are fluids? Give two examples
Give reasons
A red ink drop added to small amount of water in a glass turns the water red in some time.
For the following statement, say whether it describes a solid, a liquid or a gas.
Particles are quite close together
To identify materials of common use
Procedure – Just move around in your house – in the drawing room, sitting room, bedroom, kitchen, bathroom etc. Identify the things and All in the blanks in the table given below
| No. | Place | Name of the thing | Material used for making the thing |
| 1 | Study room | ||
| 2 | Drawing room | ||
| 3 | Kitchen | ||
| 4 | bathroom | ||
| 5 | Any other place |
Write the states of water.
Match the following.
| Srl. no. | A | B | C |
| i | Separation of visible undesirable components | Water mixed with chalk powder | Magnetic Separation |
| ii | Separation of heavier and lighter components | Sand and water | Decantation |
| iii | Separation of insoluble impurities | Iron impurities | Filtration |
| iv | Separation of magnetic components from non-magnetic components | Rice and stone | Hand-picking |
| v | Separation of solids from liquids | Husk and paddy | Winnowing |
Distinguish the properties of solid, liquid, and gas. Draw a suitable diagram.
Identify the state of matter based on the arrangement of the molecules.
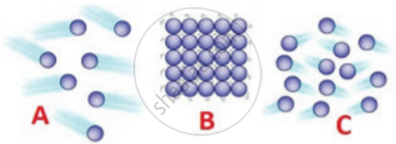
How the matter is grouped on the basis of physical states?
Give two examples of diffusion.
