Advertisements
Advertisements
Question
Explain the structure of K3[Fe(CN)6] on the basis of valence bond theory.
Explain
Advertisements
Solution
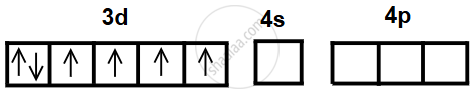
Electronic configuration of Fe = [Ar] 4s2 3d6
- The structure of K3[Fe(CN)6] is explained using Valence Bond Theory (VBT).
- The central metal ion in the complex is Fe3+.
- The atomic number of iron is 26, so its electronic configuration is [Ar] 3d6 4s2.
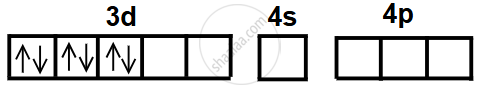
- In the +3 oxidation state, Fe3+ has the configuration 3d5, after losing two 4s electrons and one 3d electron.
- The complex contains six cyanide ions (CN−) as ligands.
- CN− is a strong field ligand and causes pairing of electrons in the 3d orbitals of Fe3+.
- This electron pairing leaves two empty 3d orbitals available for bonding.
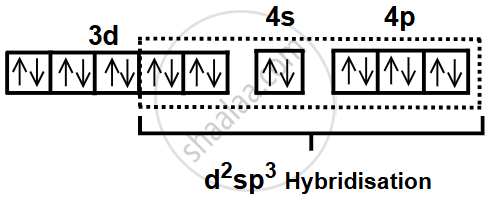
- One 4s orbital and three 4p orbitals also participate in bonding.
- These orbitals hybridize to form d2sp3 hybrid orbitals.
- Six sigma bonds are formed by the overlap of these hybrid orbitals with lone pairs from six CN− ligands.
- The resulting geometry of the complex ion [Fe(CN)6]3− is octahedral.
- Since all electrons are paired, the complex is diamagnetic.
Therefore, K₃[Fe(CN)6] is an example of an inner orbital, low-spin, octahedral complex.
shaalaa.com
Is there an error in this question or solution?
