Advertisements
Advertisements
Question
Explain the following term:
Orientation barrier.
Explain
Advertisements
Solution
To promote the production of new bonds and the breaking of existing ones, the reactant molecules must clash in a favourable orientation. Direct touch is ensured by the proper alignment. Collisions are effective because of the atoms of the molecules involved. Let's look at the following response, for instance.
\[\ce{A2 + B2 -> 2AB}\]
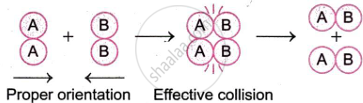

The orientation of molecules A2 and B2 leading to effective and ineffective collisions is shown in Fig.
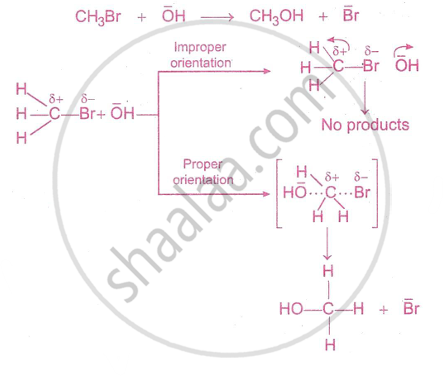
For example, the formation of methanol from bromomethane depends upon the orientation of reactant molecules, as shown in the figure below.
shaalaa.com
Is there an error in this question or solution?
