Advertisements
Advertisements
Question
Explain reactions of haloarenes with sodium metal.
Advertisements
Solution
Haloarenes undergo Wurtz-Fittig reaction and Fittig reaction on reaction with sodium metal.
- The reaction of aryl halide with alkyl halide and sodium metal in dry ether to give substituted aromatic compounds is known as Wurtz-Fittig reaction.
e.g.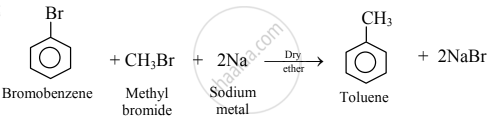
- If only aryl halide takes part in the reaction, the product is biphenyl (or diphenyl) and the reaction is known as Fittig reaction.
e.g.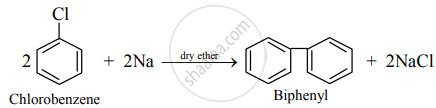
APPEARS IN
RELATED QUESTIONS
Give reason:
Reactions involving Grignard reagent must be carried out under anhydrous condition.
The reaction in which 2 molecules of chlorobenzene reacts with metallic sodium in presence of dry ether forming diphenyl is an example of ______.
Haloarenes are less reactive than alkyl halides towards nucleophilic substitution because ____________ in aryl halide.
The most suitable reagent(s) required to prepare 1-iodobutane from but-1-ene is/are ____________.
Identify the compound that gives 2, 5-dimethylhexane by reacting with sodium metal in presence of dry ether.
Identify the product of the following reaction.
\[\ce{CH3 - Br + C2H5COOAg ->[\Delta] ?}\]
The formula for the Grignard's reagent is ______.
Which of the following reaction is used to prepare toluene from bromobenzene?
Which of the following is called Wilkinson's catalyst?
Identify the product obtained in the following reaction.
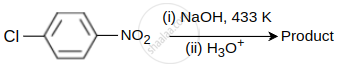
The major product obtained in the following reaction is
\[\ce{Chlorobenzene + Chlorine ->[Anhydrous][FeCl3] product (Major)}\]
Identify electron withdrawing group from following when attached to π - bond.
Which of the following alkyl halide is treated with sodium metal to obtain 2, 2, 3, 3 tetramethyl butane?
Give the following named reaction.
Fittig reaction
Write a note on Wurtz Reaction.
From Wurtz reaction one can not produce:
Write the product formed during Chlorobenzene treated with chlorine reactions.
Write the product formed Anisole treated with HI reaction.
Write the product in the following reaction:
Bromobenzene reacts with methyl bromide and sodium metal in presence of dry ether.
Write the product in the following reaction:
Chlorobenzene reacts with sodium metal in presence of dry ether.
Identify the product 'Z' in the following series of reactions.
\[\ce{\underset{}{Ethanol} ->[SOCl2][Δ] \underset{\underset{}{}}{X}->[Mg][Dry ether] \underset{\underset{}{}}{Y}->[NH3] \underset{\underset{}{}}{Z}}\]
Identify the product ‘C’ formed in the following series of reactions.
\[\ce{Bromoethane ->[Mg][Dry ether] A ->[HOH][Dry ether] B ->[Br2][UV light] C}\]
Which from following compounds is NOT obtained when ethyl chloride reacts with isopropyl chloride in presence of Na and dry ether?
Identify the CORRECT stability order of alkyl substituted alkenes from following.
