Advertisements
Advertisements
Question
Explain in detail Coulomb’s law and its various aspects.
Long Answer
Advertisements
Solution
Consider two point charges q1 and q2 at rest in vacuum, and separated by a distance of r. According to Coulomb, the force on the point charge q2 exerted by another point charge q1 is
`vec"F"_21 = "K" ("q"_1"q"_2)/"r"_2 hat"r"_12`,
where `hat"r"_12` is the unit vector directed from charge q1 to charge q2 and k is the proportionality constant.
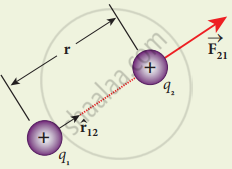
Coulomb force between two positive point charges
Important aspects of Coulomb’s law:
- Coulomb’s law states that the electrostatic force is directly proportional to the product of the magnitude of the two point charges and is inversely proportional to the square of the distance between the two point charges.
- The force on the charge q2exerted by the charge q1 always lies along the line joining the two charges. `hat"r"_21` is the unit vector pointing from charge q1 to q2 Likewise, the force on the charge q1 exerted by q2 is along – (i.e., in the direction opposite to `hat"r"_21`).
- In SI units, k = `1/(4pi ε_0)` and its value is 9 x 109 Nm2C-2. Here e0 is the permittivity of free space or vacuum and the value of ε0 = `1/(4pi ε_0)` = 8.85 x 10-12 C2 N-1 m-2
- The magnitude of the electrostatic force between two charges each of one coulomb and separated by a distance of 1 m is calculated as follows:
[F] = `(9 xx 10^9 xx 1 xx 1)/1^2 = 9 xx 10^9`N. This is a huge quantity, almost equivalent to the weight of one million ton. We never come across 1 coulomb of charge in practice. Most of the electrical phenomena in day-to-day life involve electrical charges of the order of pC (micro coulomb) or nC (nano coulomb). - In SI units, Coulomb’s law in vacuum takes the form `vec"F"_21 = 1/(4pi ε_0) ("q"_1"q"_2)/"r"_2 hat"r"_12`. sin Since ε > ε0, the force between two point charges in a medium other than vacuum is always less than that in vacuum. We define the relative permittivity for a given medium as ε = `ε/ε_0`. For vacuum or air, εr = 1 and for all other media εr > 1.
- Coulomb’s law has same structure as Newton’s law of gravitation. Both are inversely proportional to the square of the distance between the particles. The electrostatic force is directly proportional to the product of the magnitude of two point charges and gravitational force is directly proportional to the product of two masses.
- The force on a charge q1 exerted by a point charge q2 is given by `vec"F"_21 = 1/(4pi ε_0) ("q"_1"q"_2)/"r"_2 hat"r"_12`. Here `hat"r"_12` is sthe unit vector from charge q2 to q1.
But `hat"r"_21 = -hat"r"_12, vec"F"_12 = 1/(4pi ε_0) ("q"_1"q"_2)/"r"_2 (hat"r"_12)` (or) `vec"F"_12 = - vec"F"_21`
Therefore, the electrostatic force obeys Newton’s third law. - The expression for Coulomb force is true only for point charges. But the point charge is an ideal concept. However we can apply Coulomb’s law for two charged objects whose sizes are very much smaller than the distance between them. In fact, Coulomb discovered his law by considering the charged spheres in the torsion balance as point charges. The distance between the two charged spheres is much greater than the radii of the spheres.
shaalaa.com
Is there an error in this question or solution?
