Advertisements
Advertisements
Question
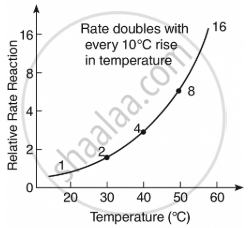
Explain graphically how the rate of a reaction changes with every 10°C rise in temperature.
Explain
Advertisements
Solution
For every 10°C rise in temperature, the rate of a chemical reaction approximately doubles.
This rule of thumb arises from the Arrhenius equation:
k = `Ae^(−E_a//(RT))`
Where:
k = rate constant
A = frequency factor
Ea = activation energy
R = gas constant
T = temperature (in Kelvin)
As temperature increases, `e^(−E_a//(RT))` increases significantly, leading to a higher value of k, and thus a faster reaction rate.
shaalaa.com
Is there an error in this question or solution?
