Advertisements
Advertisements
Question
Elements are broadly classified into metals and non-metals. State six general differences in the physical properties of metals and non-metals. State two metals and two non-metals which contradict with the general physical properties – giving reasons. State one difference in property between metalloids and noble gases.
Advertisements
Solution
Metallic elements :
- Have lustre – i.e. shine.
- Are malleable – i.e. can be beaten into sheets.
- Are ductile – i.e. can be drawn into wires.
- Are good conductors of heat and electricity.
- have high melting and boiling points.
- Have a high density.
- Contain – one type of atom – monoatomic
Non-metallic Elements :
- Do not have lustre.
- Are non-malleable – i.e. cannot be beaten into sheets.
- Are non-ductile – i.e. cannot be drawn into wires.
- Are poor conductors of heat and electricity.
- Have low melting and boiling points.
- Have a low density.
- Contain – monoatomic or diatomic atoms.
Two metals which contradict properties :
- Mercury - Mercury is liquid at room temperature whereas metals are mostly solid.
- Zinc - Zinc is Non-malleable and Non-ductile, which contradicts metal, which is malleable and ductile.
Two Non-metals are :
- Iodine - Iodine is lustrous contradicts non-metals have no lustre.
- Graphite - Graphite is a good conductor of electricity, whereas non–metals are poor conductors of electricity.
Difference between metalloids and noble gases:
Metalloids are chemically reactive whereas noble gases are chemically inert or inactive.
APPEARS IN
RELATED QUESTIONS
What are the advantages of chromatography?
Name: the Hardest Naturally Occurring Substance
Give reason: Graphite is used to make a lead of the pencils.
Fill in the blank
_________ refers to the number of atoms in the molecule of an element
Indicate whether the following statement is true or false:
Elements are made up of compounds
Tick (√) the correct alternative from the choice given for the following statement.
Sulphur and carbon are
Tick (√) the correct alternative from the choice given for the following statement.
Gold is used to make jewellery because
Give an example of two liquids that form heterogeneous mixtures.
Select the correct answer from the choice given below to complete the sentence :
Dust in air is an example of _________ mixture.
Higher Order Thinking Skills.
In the diagram given below, the circle, square and triangle represent the atoms of different elements.
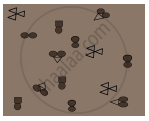
Identify all combinations that represent
- molecule of a compound
- molecule of an element consisting of two atoms
- molecule of an element consisting of three atoms
