Advertisements
Advertisements
Question
Element A belongs to group 15 and period 2 of the Periodic Table.
- Identify the element A.
- Write the formula of the compound formed when element A combines with hydrogen.
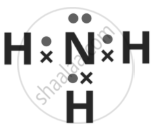
- Draw the dot (•) and cross (x) structure of the compound formed in (b) above.
Chemical Equations/Structures
Classify
Short Answer
Advertisements
Solution
(a) Element in Group 15 and Period 2 is Nitrogen (N).
(b) Nitrogen combines with hydrogen to form ammonia (NH3).
(c) Nitrogen shares three of its five valence electrons with three Hydrogen atoms, leaving one lone pair (unshared pair) of electrons.
shaalaa.com
Is there an error in this question or solution?
2025-2026 (March) Official Board Paper
