Advertisements
Advertisements
Question
Electroplating steel objects with silver involves a three-step process.
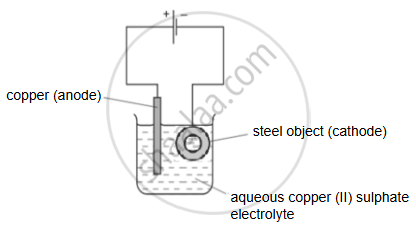
Step 1: A coating of copper is applied to the object.
Step 2: A coating of nickel is applied to the object.
Step 3: The coating of silver is applied to the object.
(a) A diagram of the apparatus used for step 1 is shown.
- The chemical process taking place on the surface of the object is \[\ce{Cu^2+_{ (aq)} + 2e- ->Cu_{(s)}}\].
What is the observation seen on the surface of the object? - Explain why the concentration of copper ions in the electrolyte remains constant throughout step 1.
(b) Give two changes which would be needed in order to coat nickel onto the object in step 2.
(c) Write down the reaction taking place at the positive electrode during step 3.
Chemical Equations/Structures
Explain
Short Answer
Advertisements
Solution
(a)
- The object will develop a coating of copper. Visually, this results in the object taking on a reddish-brown appearance due to the deposition of metallic copper. Also, there is an increase in the mass of the steel object.
- As copper ions are reduced and deposited onto the object, an equal amount of copper metal is oxidised from the anode to maintain the concentration of Cu2+ in the electrolyte solution.
(b) Two changes which would be needed in order to coat nickel onto the object in step 2 are:
- Replace the copper sulphate solution with a nickel-containing electrolyte solution, such as nickel sulphate.
- Replace the copper anode with a nickel anode.
(c) \[\ce{Ag_{(s)} -> Ag^+_{ (aq)} + e-}\]
shaalaa.com
Is there an error in this question or solution?
