Advertisements
Advertisements
Question
Draw Newman and Sawhorse projections for the eclipsed and staggered conformations of ethane. Which of these conformations is more stable and why?
Advertisements
Solution
 |
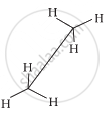 |
| Eclipsed | Staggered |
Sawhorse projection
 |
 |
| Eclipsed | Staggered |
New projection
In staggered form of ethane, the electron clouds of carbon-hydrogen bonds are as far apart as possible. Thus, there are minimum repulsive forces, minimum energy and maximum stability of the molecule. On the other hand, when the staggered form changes into the eclipsed form, the electron clouds of the carbon-hydrogen bonds come closer to each other resulting in increase in electron cloud repulsions. To check the increased repulsive forces, molecule will have to possess more energy and thus has lesser stability.
APPEARS IN
RELATED QUESTIONS
How do you account for the formation of ethane during chlorination of methane?
Rotation around carbon-carbon single bond of ethane is not completely free. Justify the statement.
Rotation of one conformer by an angle between 0° to 60° generates ______.
The dihedral angle between the hydrogen atoms of 2 methyl groups in staggered conformation of ethane is
The dihedral angle of the least stable conformer of ethane is ______.
How many conformations does ethane have?
Consider the following carbocations:


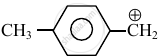
- \[\ce{CH3 - \overset{⊕}{CH2}}\]
The relative stabilities of these carbocations are such that:
