Advertisements
Advertisements
Question
Discuss the structure of PCl5.
Short Answer
Advertisements
Solution
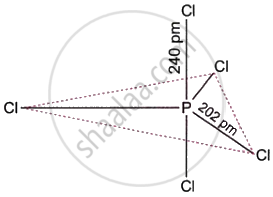
Phosphorus pentachloride (PCl5) is the most important pentahalide that is produced by group 15 elements. As seen in the figure, it features a trigonal bipyramidal structure. It contains a phosphorus atom in the middle that is sp3d-hybridized. When heated, PCl5 changes into PCl3, which is thermally more stable.
shaalaa.com
Is there an error in this question or solution?
