Advertisements
Advertisements
Question
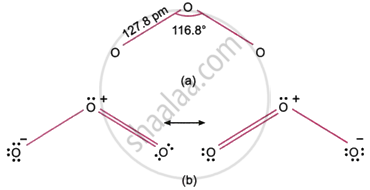
Discuss the structure of ozone.
Long Answer
Advertisements
Solution
The bond angle of the ozone molecule is 116.8°, giving it an angular or bent shape. As seen in figure (a), both of the \[\ce{O - O}\] bonds have the same length (127.8 pm, or 1.28 A). In ozone, the \[\ce{O - O}\] bond length (127.8 pm) is in between that of an \[\ce{O = O}\] double bond (110 pm) and an \[\ce{O - O}\] single bond (148 pm). It has been proposed that the ozone molecule exhibits resonance and is a resonance hybrid of the classical forms depicted in figure (b) in order to explain this phenomenon.
shaalaa.com
Is there an error in this question or solution?
