Advertisements
Advertisements
Question
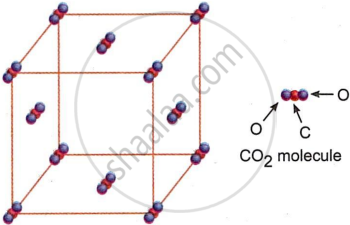
Discuss the crystal structure of dry ice.
Short Answer
Advertisements
Solution
A good example of a molecular solid is dry ice or frozen carbon dioxide. Van der Waals’ forces hold CO2 molecules together in its lattice. At 195 K (−78°C), dry ice transitions from a solid to a gaseous form due to the weakness of these forces. Fig. shows the arrangement of CO2 molecules in the crystal lattice.
shaalaa.com
Is there an error in this question or solution?
