Advertisements
Advertisements
Question
Differentiate between a physical change and a chemical change with reference to – Nature of change is temporary and reversible or permanent and irreversible.
Advertisements
Solution
| Physical change | Chemical change |
| Changes are temporary and reversible. | Change is permanent and irreversible |
APPEARS IN
RELATED QUESTIONS
Explain why burning of wood and cutting it into small piece are considered as two different types of changes.
State whether the following is physical or chemical change.
Digestion of food —
True or false
Change of seasons is a non-periodic change.
Solve this crossword by using the clues that follow-
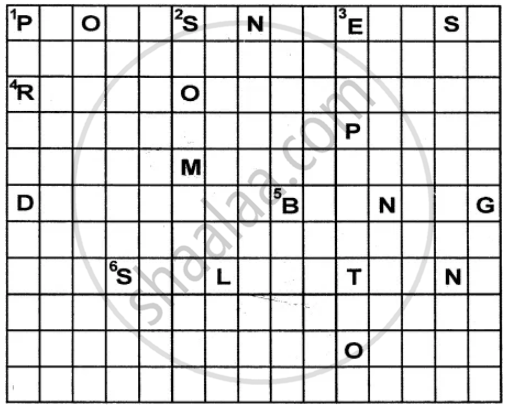
Across
1. Energy in the form of sunlight is absorbed by the green plants in this process.
4. The reddish-brown substance formed over iron in the presence of oxygen and moisture.
5. This change is permanent and irreversible.
6. The process in which a solid directly changes into gaseous state.
Down
1 . It is the change that takes place in case of swinging pendulum of a clock. –
2. Occurrence of this is a non-periodic change as well as a natural change.
3. It is a physical change.
Differentiate between a physical change and a chemical change with reference to – Energy change taking place during the respective change.
Fill in the blank with the appropriate word of the statement given below:
If a change is permanent and irreversible it is considered a _________ change.
Select the chemical changes from the following list of changes
Breaking of glass
Stretching gold coin into a ring is a ______ change.
State whether the following statement is true or false:
Stretching of rubber bands is not a physical change.
Mention some of the different conditions needed for chemical changes to occur.
