Advertisements
Advertisements
Question
Describe the principle of the following process in detail.
Chromatographic refining
Advertisements
Solution
The basis of this method is the concept that different mixture components are adsorbed on an adsorbent in different ways. The technique works particularly well for purifying elements that are only found in trace amounts and whose impurities exhibit chemical behavior that is not significantly different from that of the element that needs to be purified.
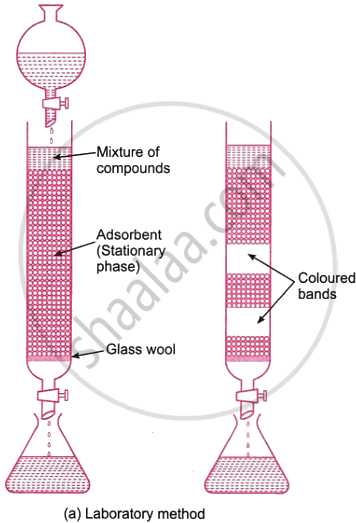
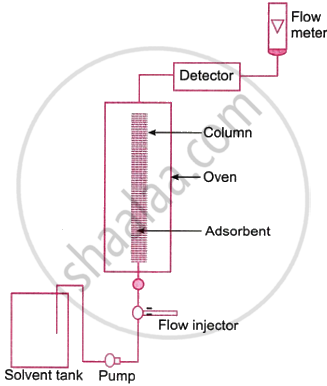
In the column chromatographic method, a glass column is filled with an adsorbent, like alumina. The combination that has to be filtered or separated is put on top of the column. To varying degrees, the mixture’s constituent parts are adsorbed in the column. An appropriate solvent, or eluent, is now created to flow through the column. First to elute is the component that is lightly adsorbed, then the component that is more highly adsorbed, and so forth. As a result, each component goes through the column individually before being gathered in different containers.
