Advertisements
Advertisements
Question
Describe the hybridisation scheme, the resultant geometry and the magnetic behaviour of [Co(NH3)6]3+.
Advertisements
Solution
Hexaamminecobalt (III) ion, [Co(NH3)6]3+:
The complex contains a cobalt (III) ion.
Atomic number of Co = 27
Electronic configuration of Co: [Ar]3d74s2

Ammonia (NH3) is a strong field ligand that causes pairing of the 3d electrons. After pairing, two 3d orbitals become vacant. These two 3d orbitals, one 4s, and three 4p orbitals mix to form d2sp3 hybrid orbitals.
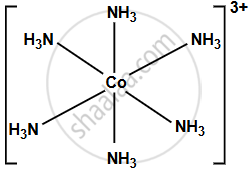
This means the six ligands (NH3) are arranged symmetrically around the central cobalt ion, forming an octahedron. They are also octahedral complexes formed by d2sp3 hybridisation and are diamagnetic in nature.
The complex [Co(NH3)6]3+ exhibits diamagnetic behavior. This means it is not attracted to a magnetic field because all of its electrons are paired. The complex has an octahedral geometry and the NH3 ligands are strong field ligands, causing the inner d-orbitals (3d) to be used in hybridization, resulting in a low-spin, or spin-paired complex.
A low-spin configuration with all paired electrons results in no unpaired electrons.
Therefore, [Co(NH3)6]3+ is diamagnetic, meaning it will not be attracted to a magnetic field.
