Advertisements
Advertisements
Question
Describe the following:
Acetylation
Chemical Equations/Structures
Short Answer
Advertisements
Solution
The substitution of active hydrogen of alcohols, phenols or amines with an acyl (–RCO) group, resulting in the formation of the corresponding ester or amides, is called acetylation. This substitution is carried out using acid chloride or acid anhydride in the presence of a base such as pyridine or dimethylaniline.
\[\ce{\underset{Acetyl Chloride}{CH3COCl} + \underset{Ethanol}{C2H5OH} ->[pyridine] \underset{Ethyl acetate}{CH3COOC2H5} + HCl}\]
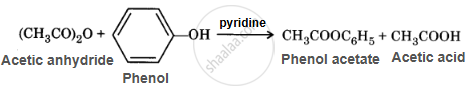
\[\ce{\underset{Acetyl Chloride}{(CH3CO)2O} + \underset{Ethyl amine}{C2H5NH2} -> \underset{N-Ethylacetamide}{CH3CONHC2H5} + HCl}\]
shaalaa.com
Is there an error in this question or solution?
Chapter 8: Aldehydes, Ketones and Carboxylic Acids - Exercises [Page 256]
