Advertisements
Advertisements
Question
Derive Laplace’s law for spherical membrane of bubble due to surface tension.
Advertisements
Solution
Consider a spherical drop as shown below. Let pi be the pressure inside the drop and po be the pressure outside it. As the drop is spherical in shape, the pressure, pi, inside the drop is greater than po, the pressure outside. Therefore, the excess pressure inside the drop is pi − po.
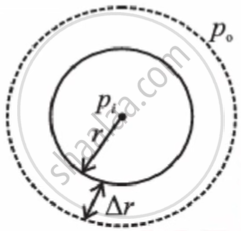
Let the radius of the drop increase from r to r + Δr, where Δr is very small, so that the pressure inside the drop remains almost constant.
Let the initial surface area of the drop be:
A1 = 4πr2
The final surface area of the drop is:
A2 = 4π(r + Δr)2
= 4π(r2 + 2rΔr + Δr2)
= 4πr2 + 8πrΔr + 4πrΔr2
As Δr is very small, πrΔr2 can be neglected.
∴ A2 = 4πr2 + 8πrΔr
Thus, the increase in surface area of the drop is:
dA = A2 − A1
= 8πrΔr
Work done in increasing the surface area by dA is stored as excess surface energy.
∴ dW = T . dA
= T(8πrΔr) ...(i)
This work done is also equal to the product of the force F, which causes an increase in the area of the drop, and the displacement Δr, which is the increase in the radius of the bubble.
∴ dW = F . Δr
The excess force is given by, (Excess pressure) × (Surface area)
dW = (pi − po) × 4πr2 × Δr ...(ii)
From equations (i) and (ii), we get,
(pi − po) × 4πr2 × Δr = T(8πrΔr)
∴ (pi − po) = `(2 r)/r`
This equation gives the excess pressure inside a drop. This is called Laplace’s law of a spherical membrane.
