Advertisements
Advertisements
Question
Curium is a radioactive element with the symbol \[\ce{^247_96{Cm}}\] named in honour of Madam Curie. The graph of number of protons vs number of neutrons for some elements are shown below:
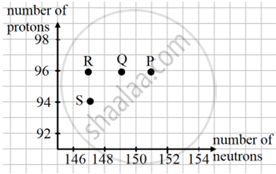
- Which point on the graph indicates the element Cm?
- Which point on the graph indicates daughter nucleus after Cm undergoes radioactive decay of 1 α followed by 2 β?
- State the mass number of the daughter nucleus.
Very Long Answer
Advertisements
Solution
Since,
Number of neutrons = Atomic mass number − Atomic number
n = A − Z
= 247 − 96
= 151
- The point P on the graph indicates the element Curium Crm.
- The point shows the daughter nuclei resulting from the radioactive decay of Cm into one alpha particle and two beta particles.
The atomic mass number and atomic number change to 243 and 94, respectively, when one alpha particle is released. The atomic mass number and atomic number 96 remain the same once 2p is released. A − Z = 243 − 96 = 147 is the number of neutrons. Therefore, R is the daughter nucleus that corresponds to 147 neutrons. - Additionally, when two beta particles are released, the mass number for the nucleus R increases to 243 and the atomic number to 96.
shaalaa.com
Is there an error in this question or solution?
2024-2025 (March) Official Board
