Advertisements
Advertisements
Question
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
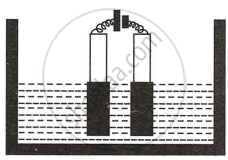
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
Answer in Brief
Advertisements
Solution
- The electrode on the left (anode) side donates electrons and hence is the oxidizing electrode.
- \[\ce{Cu - 2e- -> Cu^{2+}}\]
- The anode size steadily lowers while the cathode size gradually increases. However, the colour of the copper sulphate solution does not change.
shaalaa.com
Electrolytes
Is there an error in this question or solution?
