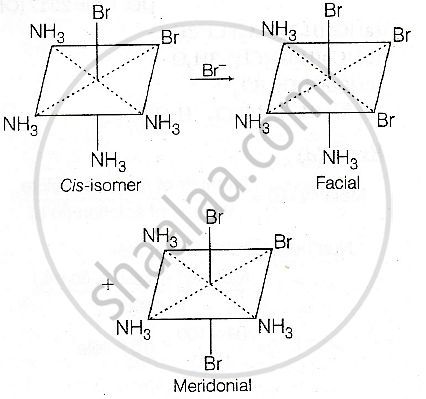
- Starting from cis-[Co(NH3)4Br2]+, replacing one NH3 by Br− can give both fac- and mer-[Co(NH3)3Br3] (two isomers).
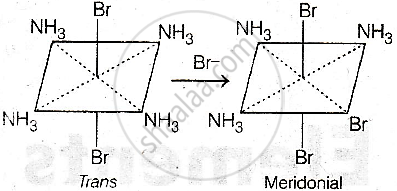
- Starting from trans-[Co(NH3)4Br2]+, the product must contain a trans Br–Br pair, so only the mer form is possible (one isomer).
Advertisements
Advertisements
Question
Consider the following reaction and statements:
\[\ce{[Co(NH3)4Br2]^+ + Br^- -> [Co(NH3)3Br3] + NH3}\]
- Two isomers are produced if the reactant complex ion is a cis-isomer.
- Two isomers are produced if the reactant complex ion is a trans-isomer.
- Only one isomer is produced if the reactant complex ion is a trans-isomer.
- Only one isomer is produced if the reactant complex ions is a cis-isomer.
The correct statements are:
Options
(I) and (II)
(I) and (III)
(III) and (IV)
(II) and (IV)
MCQ
Advertisements
Solution
(I) and (III)
Explanation:
shaalaa.com
Is there an error in this question or solution?
