Advertisements
Advertisements
Question
Consider the following figure and choose the correct option:
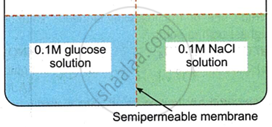
Options
There will be no movement of any solution across the membrane.
The osmotic pressure of glucose solution is equal to that of NaCl solution.
The osmotic pressure of NaCl solution is lower than that of glucose solution.
The osmotic pressure of NaCl solution is higher than that of glucose solution.
MCQ
Advertisements
Solution
The osmotic pressure of NaCl solution is higher than that of glucose solution.
Explanation:
Glucose is a non-electrolyte; it does not dissociate in solution.
Van’t Hoff factor (i) = 1
NaCl is an electrolyte; it dissociates into 2 ions (Na+ and Cl−).
Van’t Hoff factor (i) = 2
Osmotic pressure of NaCl is higher than osmotic pressure of glucose.
shaalaa.com
Is there an error in this question or solution?
