Advertisements
Advertisements
Question
Classify the following compounds based on the pattern of the carbon chain and give their structural formula:
(i) Propane
(ii) Benzene
(iii) Cyclobutane
(iv) Furan
Short/Brief Note
Advertisements
Solution
- Propane is an open chain or a cyclic compound because it contains an open chain.
CH3-CH3-CH3 [Propane]. - Benzene is a carbocyclic compound because it contains carbon atoms cyclic ring of 6 atoms.
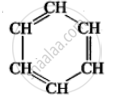
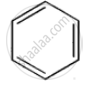 (Benzene)
(Benzene) - Cyclobutane is a carbocyclic compound.

\[\begin{array}{cc} \ce{CH2 - CH2}\\ |\phantom{........}|\\
\ce{CH2 - CH2}
\phantom{.}\\ \end{array}\]
(Cyclobutane) - Furan is a heterocyclic compound because in the cyclic chain one atom is an oxygen atom. HC – CH
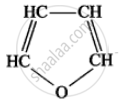
(Furan)
shaalaa.com
Classification of Organic Compounds Based on the Pattern of Carbon Chain
Is there an error in this question or solution?
