Advertisements
Advertisements
Question
\[\ce{(CH3)3 - C - CH(OH) CH3 ->[con H2SO4] X (major product)}\]
Options
(CH3)3 CCH = CH2
(CH3)2 C = C (CH3)2
CH2 = C(CH3)CH2 – CH2 – CH3
CH2 = C (CH3) – CH2 – CH2 – CH3
Advertisements
Solution
(CH3)2 C = C (CH3)2
APPEARS IN
RELATED QUESTIONS
The reaction is an example of
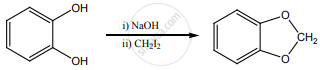
Arrange the following in the increasing order of their boiling point and give a reason for your ordering.
Butan-2-ol, Butan-1-ol, 2-methyl propane-2-ol
Can we use nucleophiles such as NH3, CH3O for the Nucleophilic substitution of alcohols?
Explain Kolbe’s reaction.
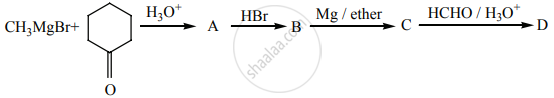
Identify A, B, C, D and write the complete equation.
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in the following method.
Acid catalysed hydration
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
What will be the product (X and A) for the following reaction.
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+] X ->[acid K2Cr2O7] A}\]
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Predict the major product, when 2-methyl but-2-ene is converted into an alcohol of the following method:
Acid catalysed hydration
