Advertisements
Advertisements
Question
Observe the following and answer the questions given below:
\[\ce{CH3 - CH3 ->[U.V. light] \overset{\bullet}{C}H3 + \overset{\bullet}{C}H3}\]
- Name the reactive intermediate produced.
- Indicate the movement of electrons by a suitable arrow to produce this intermediate.
- Comment on the stability of this intermediate produced.
Short/Brief Note
Advertisements
Solution
- The reactive intermediates produced are methyl free radicals.
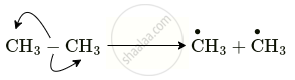
- The methyl free radicals are highly unstable neutral species formed having unpaired electrons their unstability is on account of incomplete octet unprepared election.
shaalaa.com
Is there an error in this question or solution?
