Advertisements
Advertisements
Question
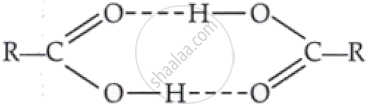
Carboxylic acids have higher boiling points than aldehydes, ketones and even alcohols of comparable molecular mass. It is due to their:
Options
more extensive association of carboxylic acid viavan der Waals force of attraction
formation of carboxylate ion
formation of intramolecular H-bonding
formation of intermolecular H-bonding
MCQ
Advertisements
Solution
formation of intermolecular H-bonding
Explanation:
Due to formation of intermolecular H-bonding in carboxylic acid, association occurs. Hence boiling point increases and become more than the boiling point of aldehydes, ketones and alcohols of comparable molecular masses.
shaalaa.com
Is there an error in this question or solution?
