Advertisements
Advertisements
Question
Carbon cannot reduce the oxides of sodium, magnesium, and aluminum to their respective metals. Why? Where are these metals placed in the reactivity series? How are these metals obtained from their ores? Take an example to explain the process of extraction along with chemical equations.
Advertisements
Solution
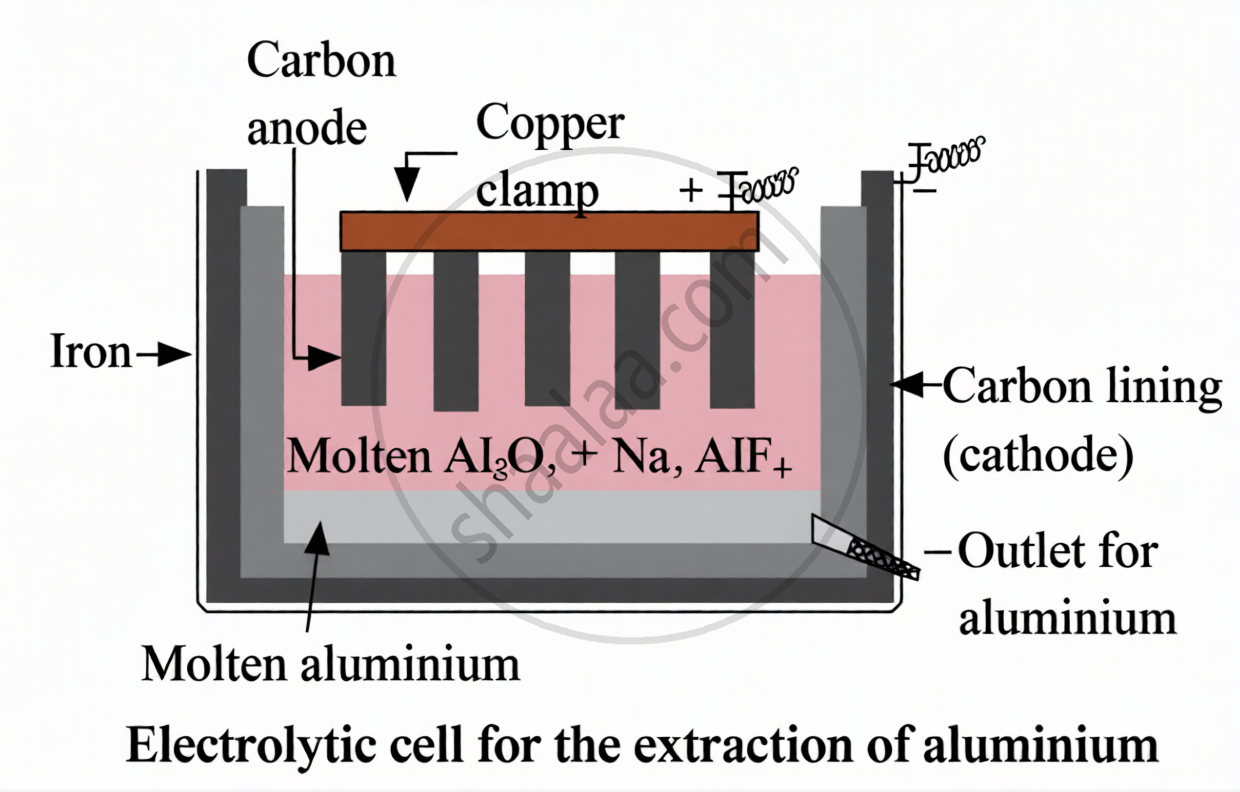
Metals like sodium, cakium, magnesium, aluminum, and others that are ranked high in the reactivity series are highly reactive. Compared to carbon, these metals are more attracted to oxygen. Consequently, it is not possible to get these metals through carbon reduction. The iron The electrolytic reduction technique is used to obtain metal for such metals.
Electrolytic reduction of aluminum: At the cathode, pure aluminum is created by electrolyzing molten aluminum oxide, while at the anode, oxygen gas is formed.
Al3+ + 3e− → Al (aluminium metal at the (−) cathode)
202− − 4e− → O2 (oxygen gas at the (+)anode)