Advertisements
Advertisements
Question
Calculate the total amount of heat energy required to convert 100 g of ice at −10℃ completely into water at 100℃. Specific heat capacity of ice = 2.1 J g-1 K-1, specific heat capacity of water = 4.2 J g-1K-1, specific latent heat of ice = 336 J g-1.
Numerical
Advertisements
Solution
Initial temperature = -10°C
Final temperature = 100°C
Total heat required to convert 100 g of ice at -10°C to water at 100°C,
Q = mCi T + mLi + mCW T + mLS
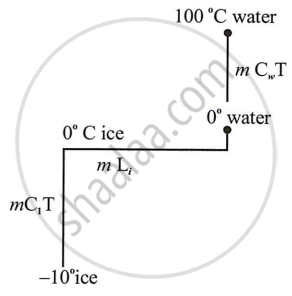
= 100 [2.1 × (0 - (-10)] + 336 + {4.2 × (100 - 0)}
= 100 `[(21/10 xx 10) + 336 + (42/10 xx 100)]`
= 100 [21 + 336 + 420]
= 100 × 777
= 7.77 × 104 J
shaalaa.com
Is there an error in this question or solution?
