Advertisements
Advertisements
Question
Calculate the radii of octahedral voids in terms of the radius of the spheres forming close-packed structures.
Advertisements
Solution
An octahedral void is generated when six nearest neighbours (atoms) are positioned in an octahedral arrangement and mutually touch. If the constituent particles are supposed to be spheres, four spheres form a plane, one above and one below it. Assume the radius of each sphere is R and the octahedral void is r.
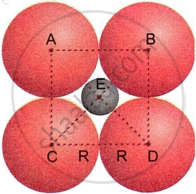
A sphere with radius r fits snugly into the octahedral void shown in the figure. The drawing shows a shaded sphere sitting in the nothingness. The spheres above and below the plane are not shown in the figure. Using the right-angled triangle ECD, we have
CD = `sqrt (CE^2 + ED^2` ...(i)
From the figure, it is clear that
CD = R + R
= 2R
EC = ED
= R + r
Putting the values in eq. (i), we have
`2R = sqrt((R + r)^2 + (R + r)^2)`
`2 R = sqrt 2 (R + r)`
`sqrt 2 R = R + r`
`r = (sqrt 2 - 1)R`
`r/R = (sqrt 2 - 1)`
`r/R = 1.414 - 1`
`r/R = 0.414`
A particle with a radius smaller than 0.414 times that of the constituent particle can only be inserted in an octahedral vacuum without disrupting the crystal lattice (close-packed structure).
