Advertisements
Advertisements
Question
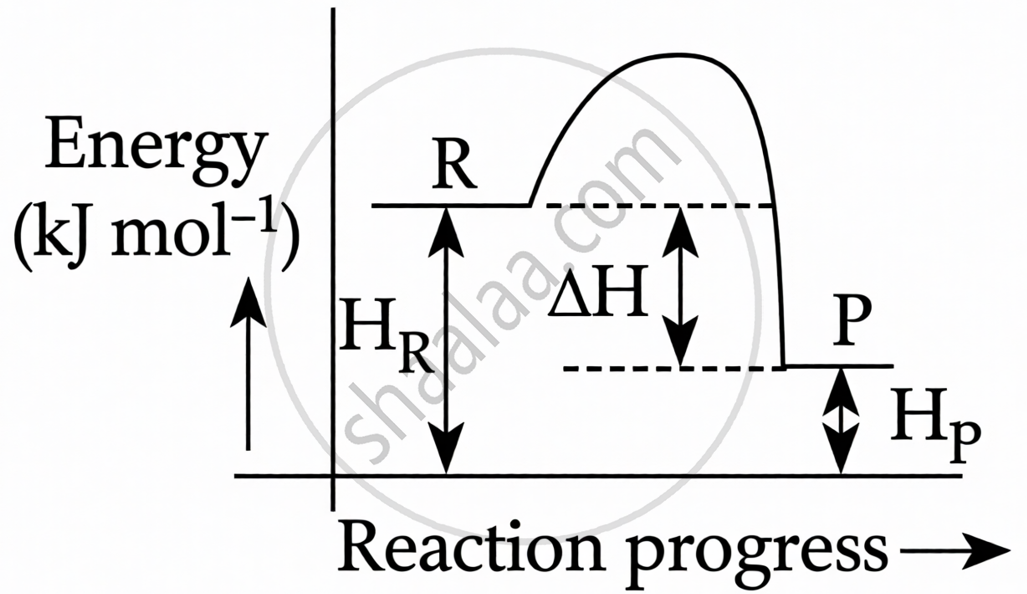
\[\ce{C(s) + 2H2(g) → CH4(g)}\]; ΔH= –74.8 kJ mol−1. Which of the following diagrams gives an accurate representation of the above reaction?
Options
MCQ
Advertisements
Solution
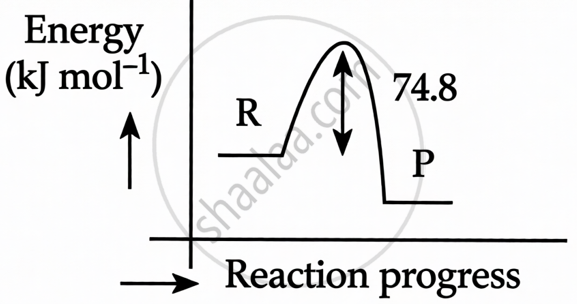
Explanation:
Exothermic Reaction: The negative sign in ΔH= –74.8 kJ mol−1 indicates that the reaction is exothermic (heat is released).
Energy Level of Products: In an exothermic reaction, the Products must have less energy than the Reactants because energy is lost to the surroundings.
HR > HP
ΔH = Hp – HR = –ve
shaalaa.com
Is there an error in this question or solution?