Advertisements
Advertisements
Question
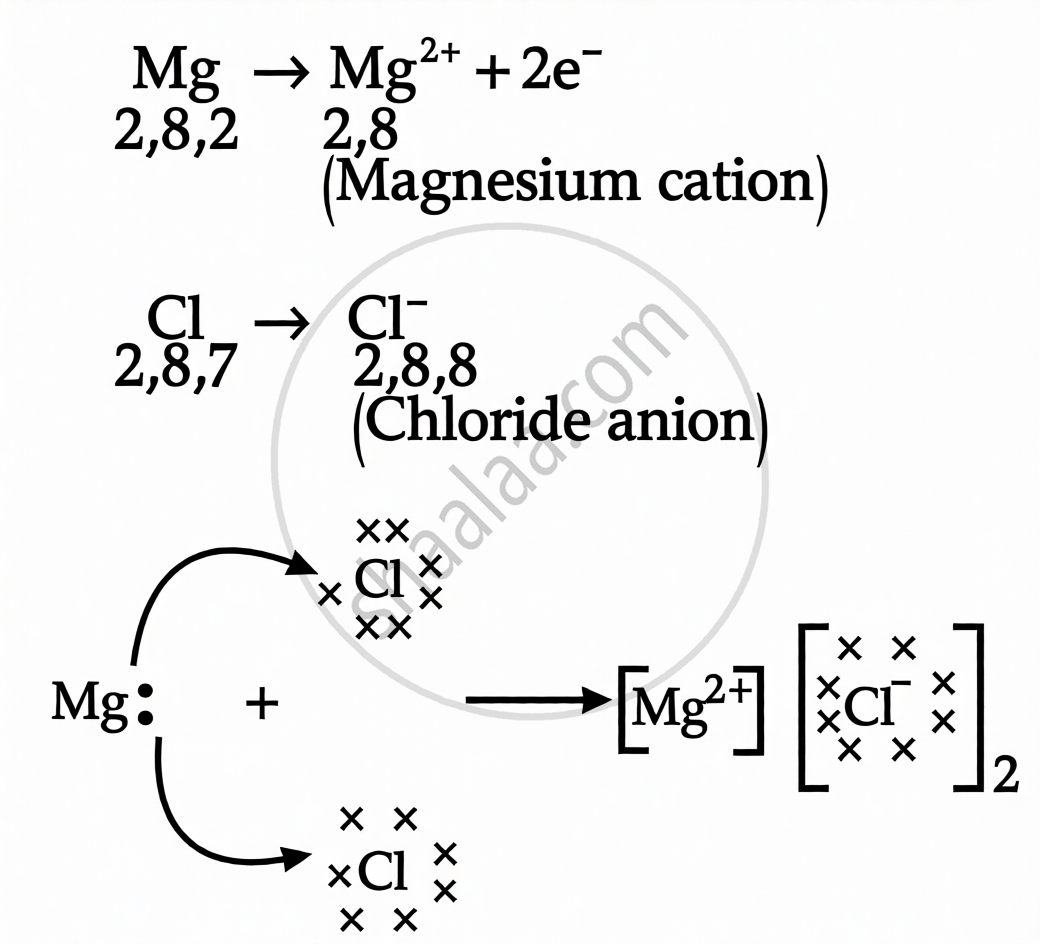
By the transfer of electrons, illustrate the formation of bond in magnesium chloride and identify the ions present in this compound.
Long Answer
Advertisements
Solution
In this case, chlorine is a nonmetal while magnesium is a metal. The magnesium cation Mg2+ is created when a magnesium atom loses two electrons to achieve the noble gas state. In a similar manner, an electron is added to a chlorine atom to complete its octet, forming the chloride anion C1-.
Two electrons are gained by two chlorine atoms when magnesium and chlorine react. Due to their opposing charges, Mg+ and Cl- are drawn to one another and are held together by powerful electrostatic forces to form MgCl2.
shaalaa.com
Is there an error in this question or solution?
2019-2020 (March) Outside Delhi set 1
