Advertisements
Advertisements
Question
Briefly discuss the observations of Hertz, Hallwachs and Lenard.
Advertisements
Solution
Hertz observation:
- In 1887, Heinrich Hertz first became successful in generating and detecting electromagnetic waves with his high voltage induction coil to cause a spark discharge between two metallic spheres.
- When a spark is formed, the charges will oscillate back and forth rapidly and electromagnetic waves are produced.
- The electromagnetic waves thus produced were detected by a detector that has a copper wire bent in the shape of a circle. Although the detection of waves is successful, there is a problem in observing the tiny spark produced in the detector.
- In order to improve the visibility of the spark, Hertz made many attempts and finally noticed an important thing that small detector spark became more vigorous when it was exposed to ultraviolet light.
- The reason for this behaviour of the spark was not known at that time. Later it was found that it is due to the photoelectric emission.
- Whenever ultraviolet light is incident on the metallic sphere, the electrons on the outer surface are emitted which caused the spark to be more vigorous.
Hallwachs’ observation:

Irradiation of ultraviolet light on
- uncharged zinc plate
- negatively charged plate
- positively charged plate
- In 1888, Wilhelm Hallwachs, a German physicist, confirmed that the strange behaviour of the spark is due to the action of ultraviolet light with his simple experiment.
- A clean circular plate of zinc is mounted on an insulating stand and is attached to a gold leaf electroscope by a wire. When the uncharged zinc plate is irradiated by ultraviolet light from an arc lamp, it becomes positively charged and the leaves will open.
- Further, if the negatively charged zinc plate is exposed to ultraviolet light, the leaves will close as the charges leaked away quickly. If the plate is positively charged, it becomes more positive upon UV rays irradiation and the leaves will open further.
- From these observations, it was concluded that negatively charged electrons were emitted from the zinc plate under the action of ultraviolet light.
Lenard’s observation:
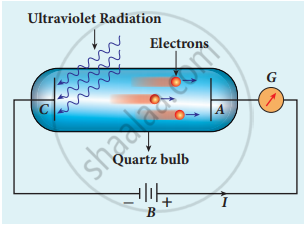
Experimental setup of Lenard
- In 1902, Lenard studied this electron emission phenomenon in detail. The apparatus consists of two metallic plates A and C placed in an evacuated quartz bulb. The galvanometer G and battery B are connected in the circuit.
- When ultraviolet light is incident on the negative plate C, an electric current flows in the circuit that is indicated by the deflection in the galvanometer. On other hand, if the positive plate is irradiated by ultraviolet light, no current is observed in the circuit.
- From these observations, it is concluded that when ultraviolet light falls on the negative plate, electrons are ejected from it which are attracted by the positive plate A. On reaching the positive plate through the evacuated bulb, the circuit is completed and the current flows in it.
- Thus, the ultraviolet light falling on the negative plate causes electron emission from the surface of the plate.
APPEARS IN
RELATED QUESTIONS
A light of wavelength 500 nm is incident on a sensitive metal plate of photoelectric work function 1.235 eV. The kinetic energy of the photoelectrons emitted is (Take h = 6.6 × 10–34 Js)
The work functions for metals A, B and C are 1.92 eV, 2.0 eV and 5.0 eV respectively. The metal/metals which will emit photoelectrons for a radiation of wavelength 4100Å is/are
How will you define threshold frequency?
What is a photocell?
Explain the effect of potential difference on photoelectric current.
Calculate the energies of the photons associated with the following radiation:
- violet light of 413 nm
- X-rays of 0.1 nm
- radio waves of 10 m
How many photons of frequency 1014 Hz will make up 19.86 J of energy?
When a 6000 Å light falls on the cathode of a photo cell, photoemission takes place. If a potential of 0.8 V is required to stop emission of electron, then determine the
- frequency of the light
- energy of the incident photon
- work function of the cathode material
- threshold frequency and
- net energy of the electron after it leaves the surface.
A 3310 Å photon liberates an electron from a material with energy 3 × 10−19 J while another 5000 Å photon ejects an electron with energy 0.972 × 10−19 J from the same material. Determine the value of Planck’s constant and the threshold wavelength of the material.
At the given point of time, the earth receives energy from the sun at 4 cal cm–2 min–1. Determine the number of photons received on the surface of the Earth per cm2 per minute. (Given: Mean wavelength of sunlight = 5500 Å)
