Advertisements
Advertisements
Question
Assertion A: Diastereomers can be easily separated by fractional distillation. Reason R: Diastereomers have different physical properties such as melting point, boiling point, solubilities in a given solvent. In the light of the above statements, choose the most appropriate answer from the options given below.
Options
Assertion is correct, reason is correct; reason is the correct explanation for assertion.
Assertion is correct, reason is correct; reason is not the correct explanation for assertion.
Assertion is correct, reason is incorrect.
Assertion is incorrect, reason is correct.
Advertisements
Solution
Assertion is correct, reason is correct; reason is not the correct explanation for assertion.
Explanation:
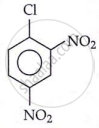
Withdrawal of electrons by -NO2 groups from ortho or para positions cause easier removal of -Cl atom due to the development of positive charge on ortho and para positions.
