Advertisements
Advertisements
Question
Arrange the following in increasing order of reactivity towards nitration
- p-xylene
- bromobenzene
- mesitylene
- nitrobenzene
- benzene
Choose the correct answer from the options given below:
Options
C < D < E < A < B
D < B < E < A < C
D < C < E < A < B
C < D < E < B < A
MCQ
Advertisements
Solution
D < B < E < A < C
Explanation:
Nitro group act as an electron withdrawing group and strongly deactivates the ring and nitration occur to that compound which contains electron donating group.
| A | B | C | D | E |
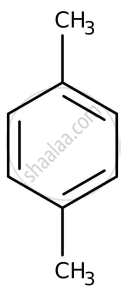 |
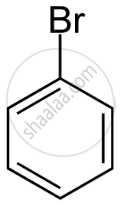 |
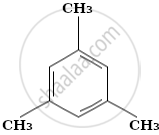 |
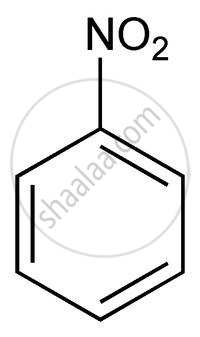 |
 |
D < B < E < A < C
shaalaa.com
Is there an error in this question or solution?
