Advertisements
Advertisements
Question
An ideal gas is pumped into a rigid container having diathermic walls so that the temperature remains constant. In a certain time interval, the pressure in the container is doubled. Is the internal energy of the contents of the container also doubled in the interval ?
Advertisements
Solution
An ideal gas is continuously being pumped into the container. Therefore, the number of moles, n are continuously increasing. In a certain interval,
Pressure, P2 = 2P1
n2 = 2n1
Thus, internal energy, U = nCvT will double as the number of moles get doubled.
APPEARS IN
RELATED QUESTIONS
In changing the state of a gas adiabatically from an equilibrium state A to another equilibrium state B, an amount of work equal to 22.3 J is done on the system. If the gas is taken from state A to B via a process in which the net heat absorbed by the system is 9.35 cal, how much is the net work done by the system in the latter case? (Take 1 cal = 4.19 J)
Two cylinders A and B of equal capacity are connected to each other via a stopcock. A contains a gas at standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. The stopcock is suddenly opened. Answer the following:
Do the intermediate states of the system (before settling to the final equilibrium state) lie on its P-V-T surface?
A cylinder containing a gas is lifted from the first floor to the second floor. What is the amount of work done on the gas? What is the amount of work done by the gas? Is the internal energy of the gas increased? Is the temperature of the gas increased?
A force F is applied on a block of mass M. The block is displaced through a distance d in the direction of the force. What is the work done by the force on the block? Does the internal energy change because of this work?
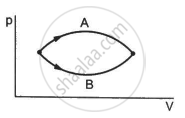
Figure shows two processes A and B on a system. Let ∆Q1 and ∆Q2 be the heat given to the system in processes A and B respectively. Then ____________ .
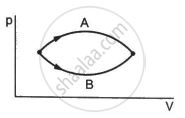
Refer to figure. Let ∆U1 and ∆U2 be the changes in internal energy of the system in the process A and B. Then _____________ .
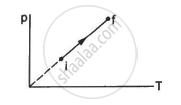
An ideal gas goes from the state i to the state f as shown in figure. The work done by the gas during the process ______________ .
A gas is taken along the path AB as shown in figure. If 70 cal of heat is extracted from the gas in the process, calculate the change in the internal energy of the system.

Which of the following is correct, when the energy is transferred to a system from its environment?
Define heat.
What is the internal energy of the system, when the amount of heat Q is added to the system and the system does not do any work during the process?
derive the relation between the change in internal energy (∆U), work is done (W), and heat (Q).
8 m3 of a gas is heated at the pressure 105 N/m2 until its volume increases by 10%. Then, the external work done by the gas is ____________.
A gas is compressed at a constant pressure of 50 N/m2 from a volume of 10 m3 to a volume of 4 m3. Energy of 100 J is then added to the gas by heating. Its internal energy is ______.
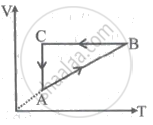
A cyclic process ABCA is shown in the V-T diagram. A process on the P-V diagram is ______.
A steam engine delivers 4.8 x 108 Jof work per minute and services 1.2 x 109 J of heat per minute from its boiler. What is the percentage efficiency of the engine?
A system releases 125 kJ of heat while 104 kJ work is done on the system. Calculate the change in internal energy.
