Advertisements
Advertisements
Question
Account for the following:
Phenol is a stronger acid than aliphatic alcohol.
Answer in Brief
Advertisements
Solution
Phenol is more acidic than alcohols due to stabilisation of phenoxide ions through resonance.
In Phenol
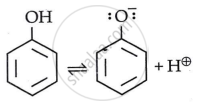

By stabilizing the phenoxide ion, the presence of an electron withdrawing group raises the acidity of phenol, while the presence of an electron releasing group decreases the acidity of phenol.
shaalaa.com
Phenols - Phenols
Is there an error in this question or solution?
