Advertisements
Advertisements
Question
According to Crystal-Field Theory, the electronic configuration of complex compound [A] is \[\ce{t^4_{2g}e^2_g}\] and that of complex compound [B] is \[\ce{t^6_{2g}e^0_g}\].
- Which of the two complex compounds, [A] or [B], is a low spin complex?
- Write the number of unpaired electrons in complex compounds [A] and [B].
- Does complex [A] have strong field ligands or weak field ligands? Give a reason.
Advertisements
Solution
i. The electronic configuration of complex [B] is \[\ce{t^6_{2g}e^0_g}\]. This shows that all six electrons are coupled, as they all occupy the lower-energy t2g orbitals. This kind of arrangement is typical of a low-spin complex, which is usually formed when strong-field ligands are present and results in a high crystal-field splitting energy (Δ0).
The electron configuration of complex [A] is \[\ce{t^4_{2g}e^2_g}\]. In this case, electrons occupy both the higher-energy e and lower-energy by orbitals, resulting in unpaired electrons. This arrangement is typical of a high-spin complex, which is typically linked to weak-field ligands that cause a reduced Δ0.
ii. Complex compound [A] has four unpaired electrons, and [B] has zero unpaired electrons.
Complex [A]: \[\ce{\mathbf{t^4_{2g}e^2_g}}\] configuration:
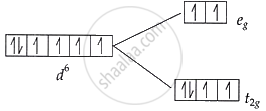
t2g level: 4 electrons → 2 unpaired electron
eg level: 2 electrons → 2 unpaired electrons
Total unpaired electrons in [A] = 4
Complex [B]: \[\ce{\mathbf{t^6_{2g}e^0_g}}\] configuration:
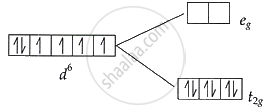
t2g level: Fully filled → 0 unpaired electrons
eg level: Empty → 0 unpaired electrons
Total unpaired electrons in [B] = 0
iii. In complex [A], the electrons are not completely paired, indicating that the ligands present produce a weak crystal field. As a result, the crystal field splitting energy (Δ0) is small, and the complex exists as a high-spin complex. In contrast, complex [B] shows complete electron pairing, which suggests the presence of strong field ligands. These ligands cause a large crystal field splitting (Δ0 > P), resulting in a low-spin complex.
