Advertisements
Advertisements
Question
- Write the mathematical expression relating the variation of rate constant of a reaction with temperature.
- How can you graphically find the activation energy of the reaction from the above expression?
Advertisements
Solution
a. The mathematical expression that relates the variation of the rate constant of a reaction with temperature is given by the Arrhenius equation:
k = `A e^(−E_a//RT)`
Where:
k = rate constant
A = pre-exponential factor or frequency factor
Ea = activation energy of the reaction
R = universal gas constant (8.314 J mol−1 K−1)
T = temperature in Kelvin
Taking the natural logarithm of both sides, the equation becomes
ln k = `ln A - E_a/(RT)`
Or in base-10 logarithm:
log k = `log A - E_a/(2.303 RT)`
This linear form is useful for plotting log k vs. `1/T` to determine Ea experimentally from the slope.
b. From the linear form:
log k = `log A - E_a/(2.303 R) * 1/T`
This resembles the equation of a straight line
y = mx + c
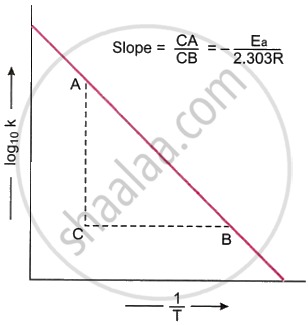
Therefore, if the values of k are plotted against `1/T`, a straight line should be obtained, and the slope of this line should be equal to `-E_a/(2.303 R)`. Therefore, in order to determine the energy of ·activation of a reaction, its rate constants at different temperatures are measured. Now, the values of log10 k against `1/T` are plotted. The curve obtained is a T straight line, as shown in Fig. The slope of the line is calculated. The activation energy of the reaction is connected to the slope of the line in the following manner.
Slope = `(CA)/(CB) = -E_a/(2.303 R)`