Advertisements
Advertisements
Question
A beam of α, β and γ rays is travelling through a certain region in space.
- Arrange them in ascending order of ionising power.
- Which of the above will pass undeviated if subjected to an electric field?
- With respect to your answer to part (b) above, what will be the change in the nucleus of an atom after such a ray is emitted.
Advertisements
Solution
- γ < β < α
- γ rays pass undeviated if subjected to an electric field.
- The nucleus of the atom has no change in the mass number and atomic number during γ emission. So, the γ rays take no mass and no electrical charge from the nucleus.
APPEARS IN
RELATED QUESTIONS
The diagram in figure shows a radioactive source S placed in a thick lead walled container. The radiations given out are allowed to pass through a magnetic field. The magnetic field (shown as ×) acts perpendicular to the plane of paper inwards. Arrows shows the paths of the radiation A, B and C.
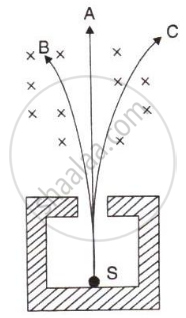
- Name the radiations labelled A, B and C.
- Explain clearly how you used the diagram to arrive at the answer in part (a).
In following Figure shows a mixed source R of alpha and beta particles in a thick lead walled container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper inwards as shown by ×.
- Show in the diagram how the particles get affected.
- Name the law used in part (a).
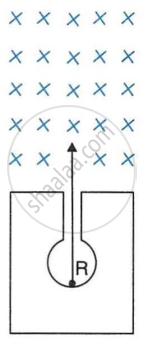
[Hint: Alpha particles will deflect to the left while beta particles to the right]
Is it possible to deflect γ - radiations in a way similar to α and β -particles, using the electric or magnetic field? Give reasons.
What is the composition of α, β and γ radiations?
Arrange the α, β, and γ radiation in ascending order of their biological damage. Give reason.
Compare the penetrating powers of α, β and γ-radiations.
Compare the ionising powers of α, β and γ radiations.
What are α-and β-radiations?
Which nuclear radiation will travel undeviated in an electric field?
Arrange the α, β and γ radiations in ascending order of their penetrating power.
