Please select a subject first
Advertisements
Advertisements
In the given reaction,
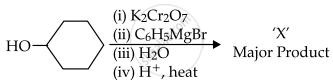
the number of sp2 hybridised carbon (s) in compound 'X' is ______.
Concept: undefined >> undefined
The OH- concentration in a mixture of 5.0 mL of 0.0504 M NH4Cl and 2 mL of 0.0210 is M NH3 solution is x × 10-6 M. The value of x is ______. (Nearest integer)
[Given Kw = 1 × 10-14 and Kb = 1.8 × 10-5]
Concept: undefined >> undefined
Advertisements
At 90°C pure water has [H3O+] = 10−6. What is the value of Kω at this temperature?
Concept: undefined >> undefined
In which of the following species S atom assumes sp3 hybrid state?
(I) (SO3)
(II) SO2
(III) H2S
(IV) S8
Concept: undefined >> undefined
If a compound on analysis was found to contain C = 18.5%, H = 1.55%, Cl = 55.04% and O = 24.81%, then its empirical formula is ______.
Concept: undefined >> undefined
Of the two solvent H2O and D2O, NaCl dissolves ______.
Concept: undefined >> undefined
In the electrolysis method of boron extraction the cathode is made of ______.
Concept: undefined >> undefined
The hybridisation of carbanion is:
Concept: undefined >> undefined
A monoprotic acid in a 0.1 M solution ionizes to 0.001%. Its ionisation constant is ______.
Concept: undefined >> undefined
Calculate pH of mixture of 400 ml of `1/200` M Ba(OH)2 400 ml of `1/50` M HCl and 200 ml of 50 water.
Concept: undefined >> undefined
400 ml of `1/200` M H2SO4, 400 ml of `1/100` M HCl and 200 ml water are mixed together, pH of the resulting solution is ______.
Concept: undefined >> undefined
Which of the following salts undergoes anionic hydrolysis?
Concept: undefined >> undefined
For the equilibrium
\[\ce{SrCl2 * 6 H2O (s) <=> SrCl2 * 2 H2O (s) + 4H2O (g)}\]
the equilibrium constant Kp = 16 × 10-12 atm4 at 1° C. If one litre of air saturated with water vapour at 1°C is exposed to a large quantity of \[\ce{SrCl2 · 2H2O(s)}\], what weight of water vapour will be absorbed? Saturated vapour pressure of water at 1°C = 7.6 torr.
Concept: undefined >> undefined
pH of 10-15 M HCl solution is ______.
Concept: undefined >> undefined
108 g of silver (molar mass 108 g mol-1) is deposited at cathode from AgNO3 (aq) solution by a certain quantity of electricity. The volume (in L) of oxygen gas produced at 273 K and 1 bar pressure from water by the same quantity of electricity is ______.
Concept: undefined >> undefined
When the sample of copper with zinc impurity is to be purified by electrolysis, the appropriate electrodes are:
Concept: undefined >> undefined
The percentage composition of carbon by a mole in methane is ______.
Concept: undefined >> undefined
116 g of a substance upon dissociation reaction, yields 7.5 g of hydrogen, 60 g of oxygen and 48.5 g of carbon. Given that the atomic masses of H, O and C are 1, 16 and 12 respectively. The data agrees with how many formulae of the following?
Concept: undefined >> undefined
In compound C, H and N atoms are present in 9 : 1 : 3.5 by weight. Molecular weight of the compound is 108. Molecular formula of the compound is ______.
Concept: undefined >> undefined
Hydrolysis of which compound will give carbolic acid?
Concept: undefined >> undefined
