Please select a subject first
Advertisements
Advertisements
250 mL of 0.5 M NaOH was added to 500 mL of 1 M HCl. The number of unreacted HCl molecules in the solution after complete reaction is ______ × 1021. (Nearest integer) (NA = 6.022 × 1023).
Concept: undefined >> undefined
A 6.50 molal solution of KOH (aq.) has a density of 1.89 g cm−3. The molarity of the solution is ______ mol dm−3. (Round off to the Nearest Integer)
[Atomic masses: K: 39.0 u; O: 16.0 u; H: 1.0 u]
Concept: undefined >> undefined
Advertisements
The number of 4f electron in the ground state electronic configuration Gd2+ is ______. (Atomic number of Gd = 64)
Concept: undefined >> undefined
Which one of the following is the correct set with respect to molecule, hybridization and shape?
Concept: undefined >> undefined
Through an aqueous solution of an unknown salt of metal M (M = 200 g/mol) a current of 1.93 A is passed for 50 min. If 4 g of metal is produced at cathode. The charge on metal ion in solution is ______.
Concept: undefined >> undefined
A current of 4 amp was passed for 2 hours through a solution of copper sulphate when 5.0 g of copper was deposited. The current efficiency is ______% (Cu = 63.5).
Concept: undefined >> undefined
A given solution of H2O2 is 30 volumes. Its concentration in terms of molarity is ______.
Concept: undefined >> undefined
A sample of Ferrous sulphide reacts with dil. H2SO4 to from H2S which contains 9% hydrogen by volume. The percentage of fee in the sample, is ______.
Concept: undefined >> undefined
The molarity of the solution prepared by dissolving 6.3 g of oxalic acid (H2C2O4 · 2 H2O) in 250 mL of water in mol L-1 is x × 10-2. The value of x is ______. (Nearest integer)
[Atomic mass: H : 1.0, C : 12.0, O : 16.0]
Concept: undefined >> undefined
The mole fraction of a solute in a 100 molal aqueous solution is ______ × 10-2. (Round off to the Nearest Integer).
[Given :Atomic masses : H : 1.0 u, O : 16.0 u]
Concept: undefined >> undefined
4 ml of pure A (d = 2.45 gm/ml) was added to 46 ml of B (d = `25.1/23` gm/ml), the molarity of a solution of A in B will be ______, if the density of the final solution is 1.8 gm/ml.
Given: Molar mass of A = 98, Molar mass of B = 46
Concept: undefined >> undefined
On passing electricity through nitrobenzene solution, it is converted into azobenzene. The mass of azobenzene is ______ mg, if the same quantity of electricity produces oxygen just sufficient to burn 96 mg of fullerene (C60)·
Concept: undefined >> undefined
Which pair of species having identical shapes?
Concept: undefined >> undefined
The correct set of four quantum numbers for the valence electrons of the rubidium atom (Z = 37) is ______.
Concept: undefined >> undefined
Which of the following is not an assumption of the kinetic theory of gases?
Concept: undefined >> undefined
By how many folds the temperature of a gas would increase when the root mean square velocity of the gas molecules in a container of fixed volume is increased from 5 × 104 cm/s to 10 × 104 cm/s?
Concept: undefined >> undefined
As the temperature is raised from 20°C to 40°C, the average kinetic energy of neon atoms changes by which factor?
Concept: undefined >> undefined
Which of the following on heating with aqueous KOH, produces acetaldehyde?
Concept: undefined >> undefined
Thiamine and pyridoxine are also known respectively as ______.
Concept: undefined >> undefined
In the given reaction,
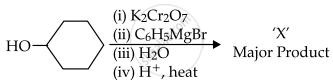
the number of sp2 hybridised carbon (s) in compound 'X' is ______.
Concept: undefined >> undefined
