Advertisements
Advertisements
Write the structures of the products when Butan-2-ol reacts with CrO3
Concept: undefined >> undefined
Write the structures of the products when Butan-2-ol reacts with SOCl2
Concept: undefined >> undefined
Advertisements
Write the structures of compounds A, B and C in the following reactions:

Concept: undefined >> undefined
Write the structures of compounds A, B and C in the following reactions
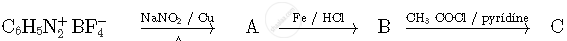
Concept: undefined >> undefined
Give reasons for the following:
CH3NH2 is more basis than C6H5NH2.
Concept: undefined >> undefined
Write the product(s) in the following reactions:

Concept: undefined >> undefined
Write the product(s) in the following reaction:

Concept: undefined >> undefined
Write the product(s) in the following reactions:

Concept: undefined >> undefined
Write the IUPAC name of the following :

Concept: undefined >> undefined
How do you convert the Ethanal to Propanone
Concept: undefined >> undefined
Write the structures of the main products in the following reactions :

Concept: undefined >> undefined
Write the names and structures of the monomers of the following polymers:
(i) Terylene
(ii) Bakelite
(iii) Buna-S
Concept: undefined >> undefined
Which one of the following is a monosaccharide:
starch, maltose, fructose, cellulose
Concept: undefined >> undefined
Calculate the emf of the following cell at 25°C :
Concept: undefined >> undefined
Give reasons for the following observations :
NH3 gas adsorbs more readily than N2 gas on the surface of charcoal.
Concept: undefined >> undefined
Give two examples of macromolecules that are chosen as drug targets
Concept: undefined >> undefined
Account for the following:
Mn2+ is more stable than Fe2+ towards oxidation to +3 state.
Concept: undefined >> undefined
Name the elements of 3d transition series that show maximum number of oxidation states. Why does this happen?
Concept: undefined >> undefined
Which transition metal of 3d series has positive E° (M2+/M) value and why?
Concept: undefined >> undefined
Out of Cr3+ and Mn3+, which is a stronger oxidising agent and why?
Concept: undefined >> undefined
