Please select a subject first
Advertisements
Advertisements
Complete the following reaction sequence.
\[\begin{array}{cc}
\ce{O}\phantom{...............................................}\\
||\phantom{...............................................}\\
\ce{CH3 - C - CH3 ->[(i) CH3MgBr][H2O] (A) ->[Na metal][Ether] (B) ->[CH3 - Br] (C)}
\end{array}\]
Concept: undefined >> undefined
Assertion: Formaldehyde is a planar molecule.
Reason: It contains sp2 hybridised carbon atom.
Concept: undefined >> undefined
Advertisements
Which of the following compound will not undergo azo coupling reaction with benzene diazonium chloride?
Concept: undefined >> undefined
The product of the following reaction is:

(i)
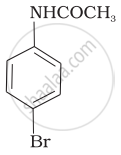
(ii)
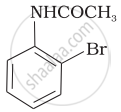
(iii)
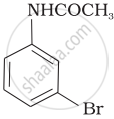
(iv)
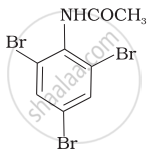
Concept: undefined >> undefined
Which of the following reactions belong to electrophilic aromatic substitution?
(i) Bromination of acetanilide
(ii) Coupling reaction of aryldiazonium salts
(iii) Diazotisation of aniline
(iv) Acylation of aniline
Concept: undefined >> undefined
Under what reaction conditions (acidic/basic), the coupling reaction of aryldiazonium chloride with aniline is carried out?
Concept: undefined >> undefined
When benzene diazonium chloride reacts with phenol, it forms a dye. This reaction is called ______.
Concept: undefined >> undefined
Iron (z=26) is highly ferromagnetic. Explain.
Concept: undefined >> undefined
What type of magnetism is shown in the following alignment of magnetic moments?

Concept: undefined >> undefined
Account for the following: Fluorine does not exhibit positive oxidation state.
Concept: undefined >> undefined
Write the major products(s) in the following:

Concept: undefined >> undefined
Name the reagents used in the following reactions:

Concept: undefined >> undefined
Predict the products of the following reactions:

Concept: undefined >> undefined
Fluorine is a stronger oxidising agent than chlorine. Why?
Concept: undefined >> undefined
What type of magnetism is shown by a substance if magnetic moments of domains are arranged in same direction?
Concept: undefined >> undefined
Why vitamin C cannot be stored in our body?
Concept: undefined >> undefined
Compare the oxidizing action of F2 and Cl2 by considering parameters such as bond dissociation enthalpy, electron gain enthalpy and hydration enthalpy.
Concept: undefined >> undefined
Account for the following :
Acidic character increases from HF to HI.
Concept: undefined >> undefined
F2 has lower bond dissociation enthalpy than Cl2. Why?
Concept: undefined >> undefined
