Advertisements
Advertisements
What should be the correct IUPAC name for diethylbromomethane?
Concept: undefined >> undefined
The reaction of toluene with chlorine in the presence of iron and in the absence of light yields ______.
Concept: undefined >> undefined
Advertisements
Which of the carbon atoms present in the molecule given below are asymmetric?
\[\begin{array}{cc}
\phantom{}\ce{HO}\phantom{.....}\ce{OH}\phantom{..}\ce{H}\phantom{.....}\ce{O}\phantom{..}\\
\phantom{..}\backslash\phantom{.....}|\phantom{....}|\phantom{.....}//\phantom{.}\\
\ce{\overset{a}{C} - \overset{b}{C} - \overset{c}{C} - \overset{d}{C}}\\
\phantom{..}//\phantom{.....}|\phantom{....}|\phantom{....}\phantom{.}\backslash\phantom{...}\\
\phantom{}\ce{O}\phantom{......}\ce{H}\phantom{...}\ce{OH}\phantom{...}\ce{H}\phantom{}\\
\end{array}\]
Concept: undefined >> undefined
Haloalkanes contain halogen atom (s) attached to the sp3 hybridised carbon atom of an alkyl group. Identify haloalkane from the following compounds.
(i) 2-Bromopentane
(ii) Vinyl chloride (chloroethene)
(iii) 2-chloroacetophenone
(iv) Trichloromethane
Concept: undefined >> undefined
Which of the following compounds can be classified as aryl halides?
(i) \[\ce{p-ClC6H4CH2CH(CH3)2}\]
(ii) \[\ce{p-CH3CHCl(C6H4)CH2CH3}\]
(iii) \[\ce{o-BrH2C - C6H4CH(CH3)CH2CH3}\]
(iv) \[\ce{C6H5 - Cl}\]
Concept: undefined >> undefined
Which of the products will be major product in the reaction given below? Explain.
\[\ce{CH3CH = CH2 + HI -> \underset{(A)}{CH3CH2CH2I} + \underset{(B)}{CH3CHICH3}}\]
Concept: undefined >> undefined
Classify the following compound as a primary, secondary and tertiary halide.
2-Bromo-2-methylpropane
Concept: undefined >> undefined
Why can aryl halides not be prepared by reaction of phenol with \[\ce{HCl}\] in the presence of \[\ce{ZnCl2}\]?
Concept: undefined >> undefined
Match the items of Column I and Column II.
| Column I | Column II | |
| (i) | SN1 reaction | (a) vic-dibromides |
| (ii) | Chemicals in fire extinguisher | (b) gem-dihalides |
| (iii) | Bromination of alkenes | (c) Racemisation |
| (iv) | Alkylidene halides | (d) Saytzeff rule |
| (v) | Elimination of HX from alkylhalide | (e) Chlorobromocarbons |
Concept: undefined >> undefined
Match the structures of compounds given in Column I with the classes of compounds given in Column II.
| Column I | Column II | |
| (i) | \[\begin{array}{cc} \ce{CH3 - CH - CH3}\\ |\phantom{..}\\ \ce{X}\phantom{..} \end{array}\] |
(a) Aryl halide |
| (ii) | \[\ce{CH2 = CH - CH2 - X}\] | (b) Alkyl halide |
| (iii) | 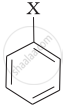 |
(c) Vinyl halide |
| (iv) | \[\ce{CH2 = CH - X}\] | (d) Allyl halide |
Concept: undefined >> undefined
Benzophenone can be obtained by:
(i) Benzoyl chloride + Benzene + \[\ce{AlCl3}\]
(ii) Benzoyl chloride + Diphenyl cadmium
(iii) Benzoyl chloride + Phenyl magnesium chloride
(iv) Benzene + Carbon monoxide + \[\ce{ZnCl2}\]
Concept: undefined >> undefined
Give the IUPAC names of the following compounds.

Concept: undefined >> undefined
Give the IUPAC names of the following compounds.

Concept: undefined >> undefined
Give the IUPAC names of the following compounds.
\[\begin{array}{cc}
\ce{CH3 - CH2 - C - CH2 - CHO}\\
||\phantom{.}\\
\ce{O}\phantom{.}
\end{array}\]
Concept: undefined >> undefined
Give the IUPAC name of the following compound.
CH3 − CH = CH − CHO
Concept: undefined >> undefined
Write IUPAC names of the following structures.
\[\begin{array}{cc}
\ce{CHO}\\
|\phantom{....}\\
\ce{CHO}\\
\end{array}\]
Concept: undefined >> undefined
Write IUPAC names of the following structures.

Concept: undefined >> undefined
Write IUPAC names of the following structures.

Concept: undefined >> undefined
Arrange the following in decreasing order of their acidic strength and give reason for your answer.
\[\ce{CH3CH2OH, CH3COOH, ClCH2COOH, FCH2COOH, C6H5CH2COOH}\]
Concept: undefined >> undefined
Which of the following is a 3° amine?
Concept: undefined >> undefined
