Advertisements
Advertisements
प्रश्न
Arrange the following in decreasing order of their acidic strength and give reason for your answer.
\[\ce{CH3CH2OH, CH3COOH, ClCH2COOH, FCH2COOH, C6H5CH2COOH}\]
Advertisements
उत्तर
The order is
\[\ce{ClCH2COOH > FCH2COOH > C6H5CH2COOH > CH3COOH > CH3CH2OH }\]
When compared to other alcohols \[\ce{CH3CH2OH}\] is the least acidic. Because of the halogen, \[\ce{FCH2COOH}\] is quite acidic. It is either an electron-donating group or an electron receiving group in acidic strength. Acidic strength increases as the number of electron withdrawing groups increases. The acidic strength of electron-donating groups diminishes.
APPEARS IN
संबंधित प्रश्न
Write the structure of 2-methylbutanal.
Name the following compound according to IUPAC system of nomenclature:
CH3CH=CHCHO
Write the IUPAC name of the following ketone or aldehyde. Wherever possible, give also the common name.

Give the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{.................}\ce{Br}\phantom{....}\ce{CH3}\phantom{.........}\ce{O}\phantom{...}\\
\phantom{................}|\phantom{......}|\phantom{............}||\phantom{..}\\
\ce{CH3 - CH2 - CH2CH - CH - CH2 - C - H}
\end{array}\]
Benzophenone can be obtained by:
(i) Benzoyl chloride + Benzene + \[\ce{AlCl3}\]
(ii) Benzoyl chloride + Diphenyl cadmium
(iii) Benzoyl chloride + Phenyl magnesium chloride
(iv) Benzene + Carbon monoxide + \[\ce{ZnCl2}\]
Give the IUPAC names of the following compounds.

Write IUPAC names of the following structures.
\[\begin{array}{cc}
\ce{CHO}\\
|\phantom{....}\\
\ce{CHO}\\
\end{array}\]
The correct IUPAC name for the given molecule should be
\[\begin{array}{cc}
\ce{H3C - \overset{H}{C} - \overset{H2}{C} - \overset{H}{C} - OH}\\
\phantom{.}|\phantom{.........}|\phantom{}\\
\phantom{...}\ce{CH3}\phantom{......}\ce{CH3}\phantom{}
\end{array}\]
IUPAC name of following compound is: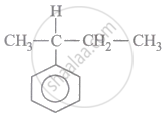
The IUPAC name of neopentane is
Complete the following:
\[\ce{CH3CN ->[1. AlH(i - Bu)2][2. H2O] 'A' ->[H2N-OH][H^+] 'B'}\]
Write IUPAC name of the following compound:
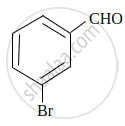
Using IUPAC norms, write the formula for the following:
Pentaamminenitrito-N-Cobalt (III)
Using IUPAC norms write the formula for the following:
Tetrahydroxidozincate (II)
Write the structure of the following compound:
Di-sec-butyl ketone
Predict the products (name and structure) in the following reaction:
\[\ce{CH3 - CONH2 ->[\Delta][dil. HCl]}\] ?
Write the structure of the following compound.
Di-sec. butyl ketone
Write the structure of the following compound.
Di-sec. butyl ketone
Write the structure of the following compound.
Di-sec. butyl ketone
