Please select a subject first
Advertisements
Advertisements
Assertion: Phenols give o- and p-nitrophenol on nitration with conc. \[\ce{HNO3}\] and \[\ce{H2SO4}\] mixture.
Reason: –OH group in phenol is o–, p– directing.
Concept: undefined >> undefined
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
Concept: undefined >> undefined
Advertisements
Explain why Lewis acid is not required in bromination of phenol?
Concept: undefined >> undefined
How can phenol be converted to aspirin?
Concept: undefined >> undefined
In Clemmensen Reduction carbonyl compound is treated with:
Concept: undefined >> undefined
Treatment of compound \[\begin{array}{cc}
\phantom{.....}\ce{O}\\
\phantom{.....}||\\
\ce{Ph - O - C - Ph}
\end{array}\] with \[\ce{NaOH}\] solution yields
(i) Phenol
(ii) Sodium phenoxide
(iii) Sodium benzoate
(iv) Benzophenone
Concept: undefined >> undefined
Arrange the following in decreasing order of their acidic strength. Give explanation for the arrangement.
\[\ce{C6H5COOH, FCH2COOH, NO2CH2COOH}\]
Concept: undefined >> undefined
Assertion: Compounds containing \[\ce{-CHO}\] group are easily oxidised to corresponding carboxylic acids.
Reason: Carboxylic acids can be reduced to alcohols by treatment with \[\ce{LiAlH4}\].
Concept: undefined >> undefined
Write down functional isomers of a carbonyl compound with molecular formula \[\ce{C3H6O}\]. Which isomer will react faster with \[\ce{HCN}\] and why? Explain the mechanism of the reaction also. Will the reaction lead to the completion with the conversion of whole reactant into product at reaction conditions? If a strong acid is added to the reaction mixture what will be the effect on concentration of the product and why?
Concept: undefined >> undefined
The letters ‘D’ or ‘L’ before the name of a stereoisomer of a compound indicate the correlation of configuration of that particular stereoisomer. This refers to their relation with one of the isomers of glyceraldehyde. Predict whether the following compound has ‘D’ or ‘L’ configuration.
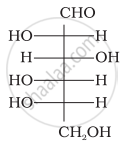
Concept: undefined >> undefined
Assertion: All naturally occurring α-aminoacids except glycine are optically active.
Reason: Most naturally occurring amino acids have L-configuration.
Concept: undefined >> undefined
A natural linear polymer of 2-methyl-1, 3-butadiene becomes hard on treatment with sulphur between 373 to 415 K and – S – S – bonds are formed between chains. Write the structure of the product of this treatment?
Concept: undefined >> undefined
Identify the type of polymer given in the following figure.
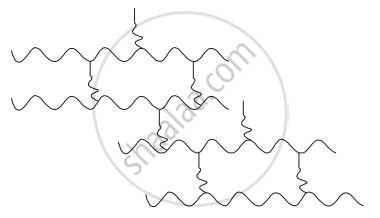
Concept: undefined >> undefined
To have practical applications why are cross links required in rubber?
Concept: undefined >> undefined
Why does cis-polyisoprene possess elastic property?
Concept: undefined >> undefined
What is the structural difference between HDP and LDP? How does the structure account for different behaviour and nature, hence the use of a polymer?
Concept: undefined >> undefined
Assertion: Network polymers are thermosetting.
Reason: Network polymers have high molecular mass.
Concept: undefined >> undefined
Which is the correct statement about birth control pills?
Concept: undefined >> undefined
The most useful classification of drugs for medicinal chemists is ______.
Concept: undefined >> undefined