Please select a subject first
Advertisements
Advertisements
What is the maximum concentration of equimolar solutions of ferrous sulphate and sodium sulphide so that when mixed in equal volumes, there is no precipitation of iron sulphide? (For iron sulphide, Ksp = 6.3 × 10–18).
Concept: undefined >> undefined
The ionization constant of nitrous acid is 4.5 × 10–4. Calculate the pH of 0.04 M sodium nitrite solution and also its degree of hydrolysis.
Concept: undefined >> undefined
Advertisements
Hydrogen bonds are formed in many compounds e.g., \[\ce{H2O, HF, NH3}\]. The boiling point of such compounds depends to a large extent on the strength of hydrogen bond and the number of hydrogen bonds. The correct decreasing order of the boiling points of above compounds is ______.
Concept: undefined >> undefined
In which of the following substances will hydrogen bond be strongest?
Concept: undefined >> undefined
Structures of molecules of two compounds are given below :
 |
 |
| (I) | (II) |
- Which of the two compounds will have intermolecular hydrogen bonding and which compound is expected to show intramolecular hydrogen bonding.
- The melting point of a compound depends on, among other things, the extent of hydrogen bonding. On this basis explain which of the above two compounds will show higher melting point.
- Solubility of compounds in water depends on power to form hydrogen bonds with water. Which of the above compounds will form hydrogen bond with water easily and be more soluble in it.
Concept: undefined >> undefined
Arrange the following in increasing order of \[\ce{pH}\].
\[\ce{KNO3 (aq), CH3COONa (aq), NH4Cl (aq), C6H5COONH4 (aq)}\]
Concept: undefined >> undefined
Consider structures I to VII and answer the question:
| I. | CH3 – CH2 – CH2 – CH2 – OH |
| II. | \[\begin{array}{cc} \ce{CH3 - CH2 - CH - CH3}\\ \phantom{.....}|\\ \phantom{.......}\ce{OH} \end{array}\] |
| III. | \[\begin{array}{cc} \phantom{...}\ce{CH3}\\ \phantom{}|\\ \ce{CH3 - C - CH3}\\ \phantom{}|\\ \phantom{..}\ce{OH} \end{array}\] |
| IV. | \[\begin{array}{cc} \ce{CH3 - CH - CH2 - OH}\\ |\phantom{........}\\ \ce{CH3}\phantom{......} \end{array}\] |
| V. | CH3 – CH2 – O – CH2 – CH3 |
| VI. | CH3 – O – CH2 – CH2 – CH3 |
| VII. | \[\begin{array}{cc} \ce{CH3 - O - CH - CH3}\\ \phantom{...}|\\ \phantom{......}\ce{CH3} \end{array}\] |
Which of the above compounds form pairs of metamers?
Concept: undefined >> undefined
The ionization constant of benzoic acid is 6.46 × 10–5 and Ksp for silver benzoate is 2.5 × 10–13. How many times is silver benzoate more soluble in a buffer of pH 3.19 compared to its solubility in pure water?
Concept: undefined >> undefined
Number of π bonds and σ bonds in the following structure is ______.
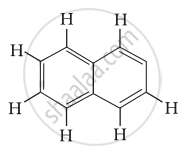
Concept: undefined >> undefined
Which molecule/ion out of the following does not contain unpaired electrons?
Concept: undefined >> undefined
Which of the following attain the linear structure?
(i) \[\ce{BeCl2}\]
(ii) \[\ce{NCO+}\]
(iii) \[\ce{NO2}\]
(iv) \[\ce{CS2}\]
Concept: undefined >> undefined
Discuss the significance/ applications of dipole moment.
Concept: undefined >> undefined
Which of the following will produce a buffer solution when mixed in equal volumes?
Concept: undefined >> undefined
On the basis of the equation \[\ce{pH}\] = – log \[\ce{[H+]}\], the \[\ce{pH}\] of 10–8 mol dm–3 solution of \[\ce{HCl}\] should be 8. However, it is observed to be less than 7.0. Explain the reason.
Concept: undefined >> undefined
Assertion (A): A solution containing a mixture of acetic acid and sodium acetate maintains a constant value of \[\ce{pH}\] on addition of small amounts of acid or alkali.
Reason (R): A solution containing a mixture of acetic acid and sodium acetate acts as a buffer solution around \[\ce{pH}\] 4.75.
Concept: undefined >> undefined
Assertion (A): An aqueous solution of ammonium acetate can act as a buffer.
Reason (R): Acetic acid is a weak acid and \[\ce{NH4OH}\] is a weak base.
Concept: undefined >> undefined
The solubility product constant of Ag2CrO4 and AgBr are 1.1 × 10–12 and 5.0 × 10–13respectively. Calculate the ratio of the molarities of their saturated solutions.
Concept: undefined >> undefined
Equal volumes of 0.002 M solutions of sodium iodate and cupric chlorate are mixed together. Will it lead to precipitation of copper iodate? (For cupric iodate Ksp = 7.4 × 10–8).
Concept: undefined >> undefined
A sparingly soluble salt gets precipitated only when the product of concentration of its ions in the solution (Qsp) becomes greater than its solubility product. If the solubility of \[\ce{BaSO4}\] in water is 8 × 10–4 mol dm–3. Calculate its solubility in 0.01 mol dm–3 of \[\ce{H2SO4}\].
Concept: undefined >> undefined
The solubility product of \[\ce{Al(OH)3}\] is 2.7 × 10–11. Calculate its solubility in gL–1 and also find out pH of this solution. (Atomic mass of \[\ce{Al = 27 u}\]).
Concept: undefined >> undefined
