SSC (English Medium)
SSC (Marathi Semi-English)
Academic Year: 2022-2023
Date & Time: 26th July 2023, 11:00 am
Duration: 2h
Advertisements
Note:
- All the questions are compulsory.
- Use of a calculator is not allowed.
- The numbers to the right of the questions indicate full marks.
- In case of MCQs [Q. No. 1(A)] only the first attempt will be evaluated and will be given credit.
- For each MCQ, the correct alternative (A), (B), (C) or (D) with the subquestion number is to be written as an answer.
For e.g.: (i) (A), (ii) (B), (iii) (C) - Scientifically correct, labelled diagrams should be drawn wherever necessary.
______ has the highest refractive index.
Air
Water
Glass
Diamond
Chapter:
The left hand side of a chemical reaction represents ______.
Product
Reactants
Catalyst
Indicator
Chapter:
In ______ block of the modern periodic table non-metals are found.
s-block
d-block
p-block
f-block
Chapter:
The chemical reaction in which two or more products are formed from a single reactant is called ______ reaction.
decomposition
combination
displacement
double displacement
Chapter: [3] Chemical Reactions and Equations [12] The Magic of Chemical Reactions
If the refractive index of glass with respect to air is `3/2`, the refractive index of air with respect to glass is ______.
`1/2`
3
`1/3`
`2/3`
Chapter:
Find the odd man out:
Camera
Telescope
Peephole in door
Microscope
Chapter:
Find the correlation:
Resistance : Ohm :: Potential difference : ______
Chapter:
Write the defect of eye from the given figure:
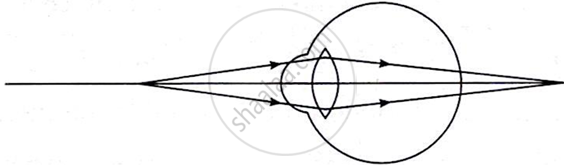
Chapter:
Advertisements
Give a scientific reason.
Tungsten metal is used to make a solenoid type coil in an electric bulb.
Chapter: [4] Effects of Electric Current [14] The Electric Spark
Give a scientific reason.
Simple microscope is used for watch repairs.
Chapter: [7] Lenses [16] Wonders of Light 1
Write a scientific reason.
Metallic character goes on decreasing while going from left to right in a period.
Chapter: [2] Periodic Classification of Elements
Write the IUPAC name of the following structural formula:
\[\begin{array}{cc}
\ce{CH3 - CH - CH3}\\
|\phantom{..}\\
\ce{OH}\\
\end{array}\]
Chapter:
Write the IUPAC name of the following structural formula:
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH2 - CH - CH3}\\
\phantom{............}|\\
\phantom{............}\ce{Cl}\\
\end{array}\]
Chapter:
An iron ball of mass 5 kg is released from a height of 125 m and falls freely to the ground. Assuming that the value of g is 10 m/s2, calculate time taken by the ball to reach the ground.
Chapter:
Draw the image formed by convex lens, if object is placed at 2F1.
Chapter:
Why does the apparent position of stars keep changing a bit?
Chapter:
Advertisements
Identify the process given below and accordingly draw neat labelled diagram:
A molten mixture of alumina (melting point > 2000°C) is done in a steel tank. The tank has a graphite lining on the inner side. The lining does the work of cathode. A set of graphite rods dipped in the molten electrolyte works as anode. Cryolite (Na3AlF6) and flurospar (CaF2) are added in the mixture to lower its melting point upto 1000°C.
Chapter:
With reference to the given diagram, answer the following questions:
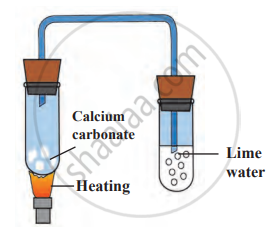
- Give type of chemical reaction.
- Give the names of reactants and products.
- Write down the balanced chemical equation.
Chapter:
What is meant by "electric power"?
Chapter: [14] The Electric Spark
Derive the unit of electric power from the given equations:
P = V × `square`
P = `square` × ampere
= 1 volt × 1`square` = `(1 "J")/(1 "C") xx (1 "C")/(1 "s")`
∴ P = `(1 "J")/square` = W(Watt).
Chapter:
The electronic configuration of an element X is 2, 8, 8, 2.
- What is the atomic number of element X?
- To which group does this element belong?
- In which period does this element lie?
Chapter:
What is the contribution of India in space technology?
Chapter:
Observe the given diagram and answer the following questions:

- What is refraction of light?
- Name the emergent ray.
- Which two angles are equal?
Chapter:
Observe the given diagram and answer the following questions:
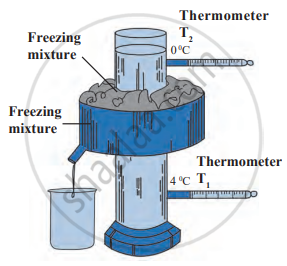
- What is the name of the given apparatus?
- Which phenomenon is studied with the help of this apparatus?
- Which are the final temperatures in thermometers T1 and T2?
- At what temperature the density of water maximum?
- Give one example of the above phenomenon in nature.
Chapter:
Observe and write the answers to the questions given below:
| I. \[\begin{array}{cc} \ce{H}\phantom{...}\ce{H}\\ |\phantom{....}|\\ \ce{H - C - C - H}\\ |\phantom{....}|\\ \ce{H}\phantom{...}\ce{H}\\ \end{array}\] |
II. \[\begin{array}{cc} \ce{H}\phantom{.........}\ce{H}\\ \backslash\phantom{.......}/\\ \ce{\phantom{.}C \phantom{.}= \phantom{.} C\phantom{.}}\phantom{.}\\ /\phantom{.......}\backslash\\ \ce{H}\phantom{.........}\ce{H}\\ \end{array}\] |
- Write the names of compound I and II.
- Draw electronic-dot structure for I and II.
- Which one of the above structures is saturated compound and unsaturated compound?
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
Maharashtra State Board previous year question papers 10th Standard Science and Technology 1 with solutions 2022 - 2023
Previous year Question paper for Maharashtra State Board 10th Standard -2023 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science and Technology 1, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of Maharashtra State Board 10th Standard.
How Maharashtra State Board 10th Standard Question Paper solutions Help Students ?
• Question paper solutions for Science and Technology 1 will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
